Back
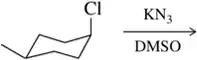
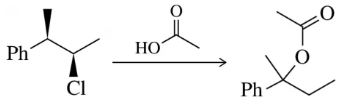
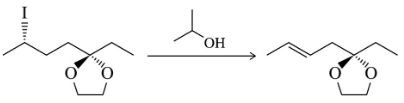
BackProblem 31a
Predict the product of the substitution reactions, paying attention to the stereochemical outcome.
(a)
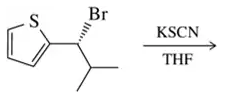
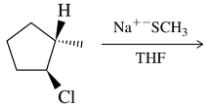
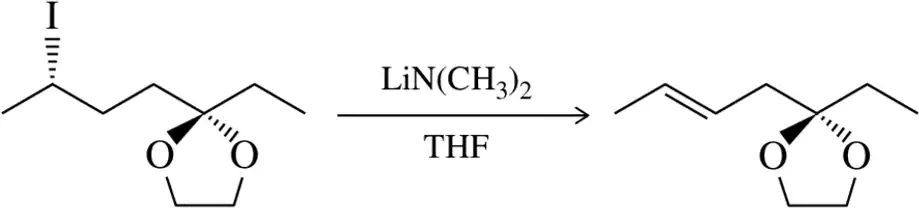
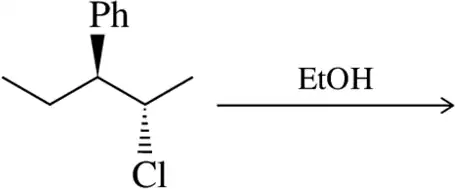
Problem 31b
Predict the product of the substitution reactions, paying attention to the stereochemical outcome.
(b)
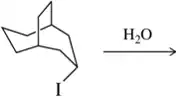
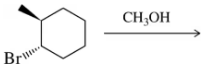
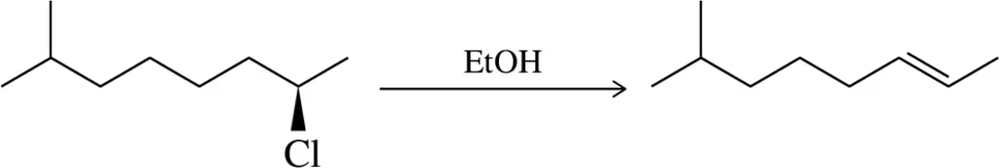
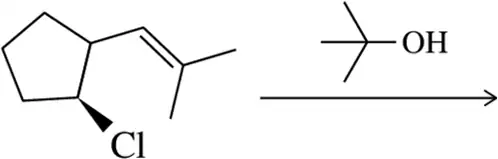
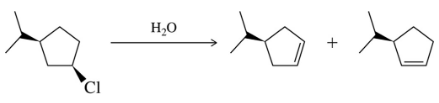
Problem 31d
Predict the product of the substitution reactions, paying attention to the stereochemical outcome.
(d)
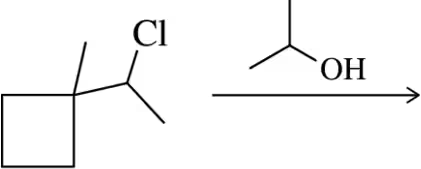
Problem 32a
Provide a mechanism for the following SN1 reactions that feature a rearrangement.
(a)
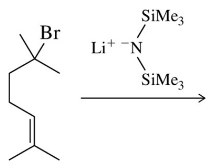
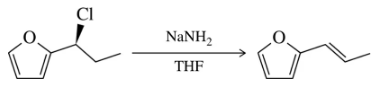
Problem 32b
Provide a mechanism for the following SN1 reactions that feature a rearrangement.
(b)
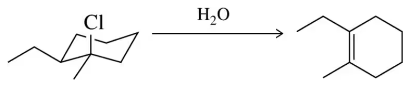
Problem 33a
Predict the product of the following substitution reactions, making sure to note whether a rearrangement should occur.
(a)
Problem 33b
Predict the product of the following substitution reactions, making sure to note whether a rearrangement should occur.
(b)
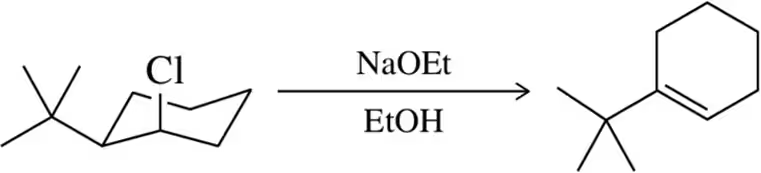
Problem 33c
Predict the product of the following substitution reactions, making sure to note whether a rearrangement should occur.
(c)
Problem 33d
Predict the product of the following substitution reactions, making sure to note whether a rearrangement should occur.
(d)
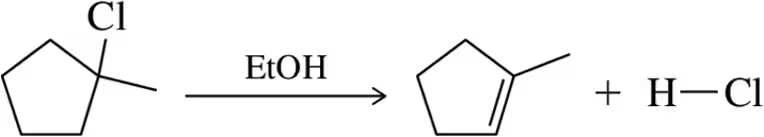
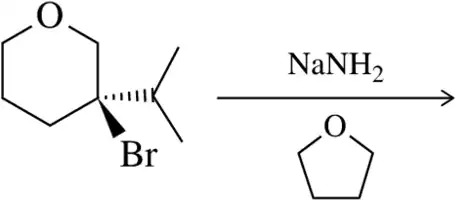
Problem 34a
Identify the bonds that break and form in the following elimination reactions.
(a)
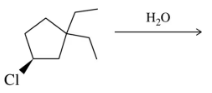
Problem 34b
Identify the bonds that break and form in the following elimination reactions.
(b)
Problem 35
When 2-bromopropane is treated with sodium ethoxide, propene is produced. What molecule is lost from 2-bromopropane in this process?
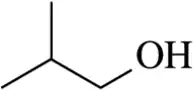
Problem 36a
(a) How would you convert propene to 2-bromopropane?
Problem 37a
Practice your electron-pushing skills by drawing a mechanism for the following E2 reactions.
(a)
Problem 37b
Practice your electron-pushing skills by drawing a mechanism for the following E2 reactions.
(b)
Problem 38a
Practice your electron-pushing skills by drawing a mechanism for the following E1 reactions.
(a)
Problem 38b
Practice your electron-pushing skills by drawing a mechanism for the following E1 reactions.
(b)
Problem 38c
Practice your electron-pushing skills by drawing a mechanism for the following E1 reactions.
(c)
Problem 39a
Would you expect the following bases to favor E1 or E2 elimination?
(a)
Problem 39c
Would you expect the following bases to favor E1 or E2 elimination?
(c)
Problem 39d
Would you expect the following bases to favor E1 or E2 elimination?
(d) H2O
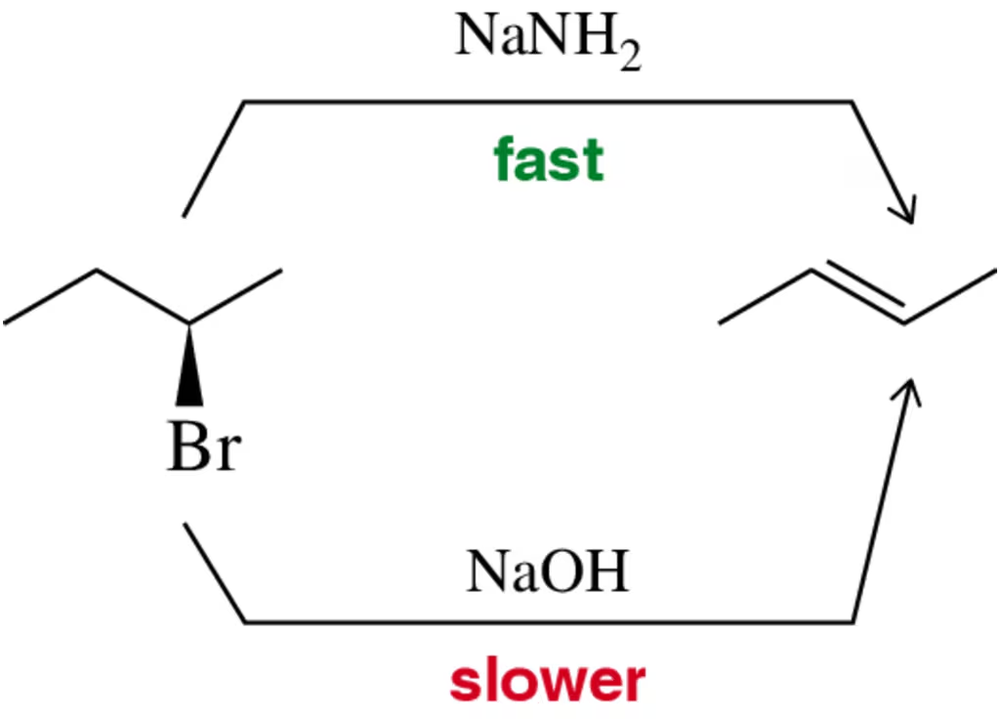
Problem 40
Justify on a reaction coordinate diagram the fact that a strong base like sodium amide (NaNH2) results in a faster E2 elimination than does sodium hydroxide (NaOH).
Problem 41a
Given the reactants shown, what type of elimination would you expect to occur?
(a)
Problem 41b
Given the reactants shown, what type of elimination would you expect to occur?
(b)
Problem 41c
Given the reactants shown, what type of elimination would you expect to occur?
(c)
Problem 41d
Given the reactants shown, what type of elimination would you expect to occur?
(d)
Problem 42a
Show a mechanism for the following elimination reactions. Label the mechanism as E1 or E2.
(a)
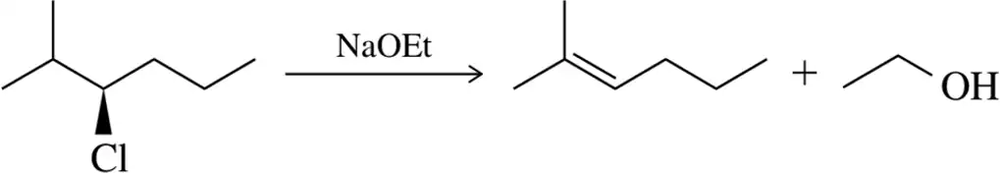
Problem 42c
Show a mechanism for the following elimination reactions. Label the mechanism as E1 or E2.
(c)
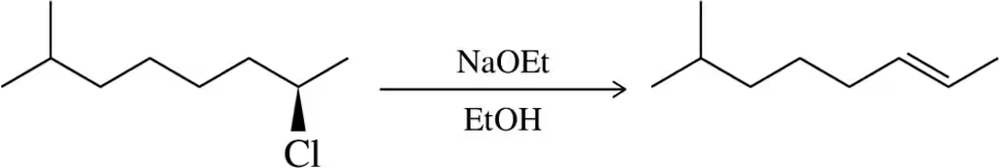
Problem 42d
Show a mechanism for the following elimination reactions. Label the mechanism as E1 or E2.
(d)
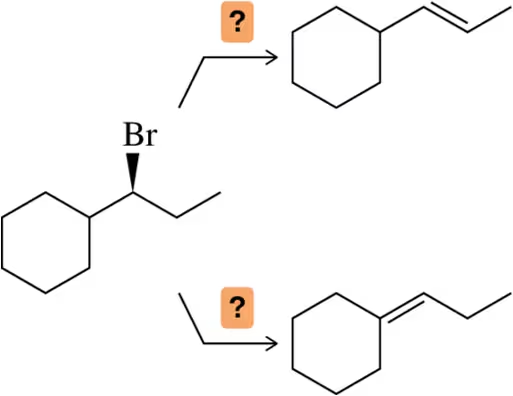
Problem 43a
Suggest an appropriate base to synthesize the alkene as the major product from the starting haloalkane.
(a)