 Back
BackProblem 26b
You are asked to prepare a pH = 4.00 buffer starting from 1.50 L of 0.0200 M solution of benzoic acid 1C6H5COOH2 and any amount you need of sodium benzoate 1C6H5COONa2. (b) How many grams of sodium benzoate should be added to prepare the buffer? Neglect the small volume change that occurs when the sodium benzoate is added.
Problem 27a
A buffer contains 0.10 mol of acetic acid and 0.13 mol of sodium acetate in 1.00 L. (a) What is the pH of this buffer?
Problem 27b
A buffer contains 0.10 mol of acetic acid and 0.13 mol of sodium acetate in 1.00 L. b. What is the pH of the buffer after the addition of 0.020 mol of KOH?
Problem 27c
A buffer contains 0.10 mol of acetic acid and 0.13 mol of sodium acetate in 1.00 L. c. What is the pH of the buffer after the addition of 0.020 mol of HNO3?
- A buffer contains 0.15 mol of propionic acid (C2H5COOH) and 0.10 mol of sodium propionate (C2H5COONa) in 1.20 L. (a) What is the pH of this buffer? (b) What is the pH of the buffer after the addition of 0.01 mol of NaOH? (c) What is the pH of the buffer after the addition of 0.01 mol of HI?
Problem 28
Problem 29a
(a) What is the ratio of HCO3- to H2CO3 in blood of pH 7.4?
Problem 29b
(b) What is the ratio of HCO3- to H2CO3 in an exhausted marathon runner whose blood pH is 7.1?
Problem 31a
You have to prepare a pH = 3.50 buffer, and you have the following 0.10 M solutions available: HCOOH, CH3COOH, H3PO4, HCOONa, CH3COONa, and NaH2PO4. Which solutions would you use?
Problem 31b
You have to prepare a pH = 3.50 buffer, and you have the following 0.10 M solutions available: HCOOH, CH3COOH, H3PO4, HCOONa, CH3COONa, and NaH2PO4. How many milliliters of each solution would you use to make approximately 1 L of the buffer?
Problem 32a
You have to prepare a pH = 5.00 buffer, and you have the following 0.10 M solutions available: HCOOH, HCOONa, CH3COOH, CH3COONa, HCN, and NaCN. Which solutions would you use?
Problem 32b
You have to prepare a pH = 5.00 buffer, and you have the following 0.10 M solutions available: HCOOH, HCOONa, CH3COOH, CH3COONa, HCN, and NaCN. How many milliliters of each solution would you use to make approximately 1 L of the buffer?
Problem 33d
The accompanying graph shows the titration curves for two monoprotic acids. (d) Estimate the pKa of the weak acid.

Problem 34a
Compare the titration of a strong, monoprotic acid with a strong base to the titration of a weak, monoprotic acid with a strong base. Assume the strong and weak acid solutions initially have the same concentrations. Indicate whether the following statements are true or false. (a) More base is required to reach the equivalence point for the strong acid than the weak acid.
Problem 35a
The samples of nitric and acetic acids shown here are both titrated with a 0.100 M solution of NaOH(aq).
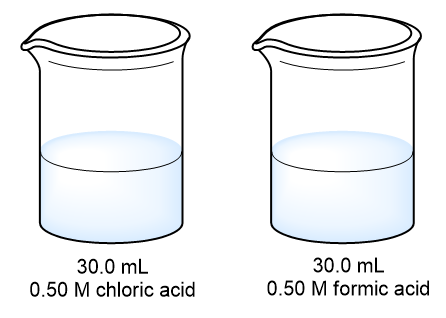
Determine whether each of the following statements concerning these titrations is true or false. (a) A larger volume of NaOH1aq2 is needed to reach the equivalence point in the titration of HNO3.
Problem 36b
Determine whether each of the following statements concerning the titrations in Problem 17.35 is true or false. b. Both titration curves will be essentially the same after passing the equivalence point.
Problem 37a,c
Predict whether the equivalence point of each of the following titrations is below, above, or at pH 7: (a) NaHCO3 titrated with NaOH (c) KOH titrated with HBr.
Problem 37b
Predict whether the equivalence point of each of the following titrations is below, above, or at pH 7: (b) NH3 titrated with HCl.
Problem 38a
Predict whether the equivalence point of each of the following titrations is below, above, or at pH 7: (a) formic acid titrated with NaOH.
Problem 38b
Predict whether the equivalence point of each of the following titrations is below, above, or at pH 7: (b) calcium hydroxide titrated with perchloric acid.
Problem 38c
Predict whether the equivalence point of each of the following titrations is below, above, or at pH 7: (c) pyridine titrated with nitric acid.
Problem 40a
Assume that 30.0 mL of a 0.10 M solution of a weak base B that accepts one proton is titrated with a 0.10 M solution of the monoprotic strong acid HA. (a) How many moles of HA have been added at the equivalence point?
Problem 40b
Assume that 30.0 mL of a 0.10 M solution of a weak base B that accepts one proton is titrated with a 0.10 M solution of the monoprotic strong acid HA. (b) What is the predominant form of B at the equivalence point?
Problem 40c
Assume that 30.0 mL of a 0.10 M solution of a weak base B that accepts one proton is titrated with a 0.10 M solution of the monoprotic strong acid HA. (c) Is the pH 7, less than 7, or more than 7 at the equivalence point?
Problem 41b
How many milliliters of 0.0850 M NaOH are required to titrate each of the following solutions to the equivalence point: (b) 35.0 mL of 0.0850 M CH3COOH?
Problem 41c
How many milliliters of 0.0850 M NaOH are required to titrate each of the following solutions to the equivalence point: (c) 50.0 mL of a solution that contains 1.85 g of HCl per liter?
Problem 42a,c
How many milliliters of 0.105 M HCl are needed to titrate each of the following solutions to the equivalence point: (a) 45.0 mL of 0.0950 M NaOH? (c) 125.0 mL of a solution that contains 1.35 g of NaOH per liter?
Problem 42b
How many milliliters of 0.105 M HCl are needed to titrate each of the following solutions to the equivalence point: (b) 22.5 mL of 0.118 M NH3?
Problem 43a,b
A 20.0-mL sample of 0.200 M HBr solution is titrated with 0.200 M NaOH solution. Calculate the pH of the solution after the following volumes of base have been added:
(a) 15.0 mL
(b) 19.9 mL.
Problem 43c
A 20.0-mL sample of 0.200 M HBr solution is titrated with 0.200 M NaOH solution. Calculate the pH of the solution after the following volumes of base have been added:
(c) 20.0 mL.
Problem 43e
A 20.0-mL sample of 0.200 M HBr solution is titrated with 0.200 M NaOH solution. Calculate the pH of the solution after the following volumes of base have been added: (e) 35.0 mL.
