Textbook Question
Ethyl acetate, CH3CO2CH2CH3, is commonly used as a solvent and nail-polish remover. Look at the following electrostatic potential map of ethyl acetate, and explain the observed polarity.
1
views

 McMurry 8th Edition
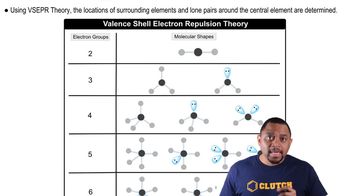
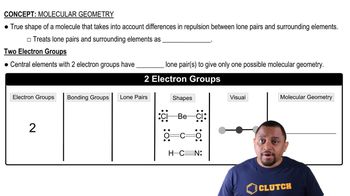
McMurry 8th Edition Ch.8 - Covalent Compounds: Bonding Theories and Molecular Structure
Ch.8 - Covalent Compounds: Bonding Theories and Molecular Structure Problem 39
Problem 39 Verified step by step guidance
Verified step by step guidance

Ethyl acetate, CH3CO2CH2CH3, is commonly used as a solvent and nail-polish remover. Look at the following electrostatic potential map of ethyl acetate, and explain the observed polarity.
Two dichloroethylene molecules with the same chemical formula 1C2H2Cl22, but different arrangements of atoms are shown. (b) Which form of dichloroethylene has a dipole moment of 2.39 D, and which has a dipole moment of 0.00 D?