 Back
BackProblem 87b
Suppose a scientist repeats the Millikan oil-drop experiment but reports the charges on the drops using an unusual (and imaginary) unit called the warmomb (wa). The scientist obtains the following data for four of the drops: Droplet Calculated Charge (wa) A 3.84⨉10−8 B 4.80⨉10−8 C 2.88⨉10−8 D 8.64⨉10−8 (b) From these data, what is the best choice for the charge of the electron in warmombs?
Problem 87c
Suppose a scientist repeats the Millikan oil-drop experiment but reports the charges on the drops using an unusual (and imaginary) unit called the warmomb (wa). The scientist obtains the following data for four of the drops: Droplet Calculated Charge (wa) A 3.84⨉10−8 B 4.80⨉10−8 C 2.88⨉10−8 D 8.64⨉10−8 (c) Based on your answer to part (b), how many electrons are there on each of the droplets?
Problem 87d
Suppose a scientist repeats the Millikan oil-drop experiment but reports the charges on the drops using an unusual (and imaginary) unit called the warmomb (wa). The scientist obtains the following data for four of the drops: Droplet Calculated Charge (wa) A 3.84⨉10−8 B 4.80⨉10−8 C 2.88⨉10−8 D 8.64⨉10−8 (d) What is the conversion factor between warmombs and coulombs?
Problem 88a
The natural abundance of 3He is 0.000137%. (a) How many protons, neutrons, and electrons are in an atom of 3He?
Problem 88b
The natural abundance of 3He is 0.000137%. (b) Based on the sum of the masses of their subatomic particles, which is expected to be more massive, an atom of 3He or an atom of 3H (which is also called tritium)?
Problem 88c
The natural abundance of 3He is 0.000137%. (c) Based on your answer to part (b), what would need to be the precision of a mass spectrometer that is able to differentiate between peaks that are due to 3He+ and 3H+?
- A cube of gold that is 1.00 cm on a side has a mass of 19.3 g. A single gold atom has a mass of 197.0 u. (c) What assumptions did you make in arriving at your answer for part (b)?
Problem 89
Problem 89a
A cube of gold that is 1.00 cm on a side has a mass of 19.3 g. A single gold atom has a mass of 197.0 u. (a) How many gold atoms are in the cube?
Problem 89b
A cube of gold that is 1.00 cm on a side has a mass of 19.3 g. A single gold atom has a mass of 197.0 u. (b) From the information given, estimate the diameter in Å of a single gold atom.
Problem 90a
The diameter of a rubidium atom is 495 pm We will consider two different ways of placing the atoms on a surface. In arrangement A, all the atoms are lined up with one another to form a square grid. Arrangement B is called a close-packed arrangement because the atoms sit in the 'depressions' formed by the previous row of atoms: (a) Using arrangement A, how many Rb atoms could be placed on a square surface that is 1.0 cm on a side?
Problem 90b
(b) How many molecules of C13H18O2 are in this tablet?
Problem 90c2
"The diameter of a rubidium atom is 495 pm We will consider two different ways of placing the atoms on a surface. In arrangement A, all the atoms are lined up with one another to form a square grid. Arrangement B is called a close-packed arrangement because the atoms sit in the 'depressions' formed by the previous row of atoms:

(c) If extended to three dimensions, which arrangement would lead to a greater density for Rb metal?"
Problem 90c1
"The diameter of a rubidium atom is 495 pm We will consider two different ways of placing the atoms on a surface. In arrangement A, all the atoms are lined up with one another to form a square grid. Arrangement B is called a close-packed arrangement because the atoms sit in the 'depressions' formed by the previous row of atoms:
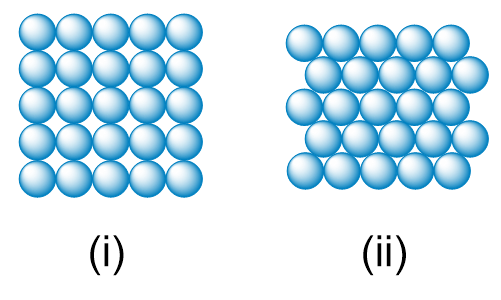
(c) By what factor has the number of atoms on the surface increased in going to arrangement B from arrangement A?
- Very small semiconductor crystals, composed of approximately 1000 to 10,000 atoms, are called quantum dots. Quantum dots made of the semiconductor CdSe are now being used in electronic reader and tablet displays because they emit light efficiently and in multiple colors, depending on dot size. The density of CdSe is 5.82 g/cm3. (b) CdSe quantum dots that are 2.5 nm in diameter emit blue light upon stimulation. Assuming that the dot is a perfect sphere and that the empty space in the dot can be neglected, calculate how many Cd atoms are in one quantum dot of this size.
Problem 91
Problem 91a
(a) Assuming the dimensions of the nucleus and atom shown in Figure 2.10, what fraction of the volume of the atom is taken up by the nucleus?
Problem 91b
(b) Using the mass of the proton from Table 2.1 and assuming its diameter is 1.0 * 10-15 m, calculate the density of a proton in g>cm3.
Problem 92a
Identify the element represented by each of the following symbols and give the number of protons and neutrons in each: (a) 7433X
Problem 92b,c,d
Identify the element represented by each of the following symbols and give the number of protons and neutrons in each: (b) 12753X (c) 8636X (d) 6730X
Problem 93a
The nucleus of 6Li is a powerful absorber of neutrons. It exists in the naturally occurring metal to the extent of 7.5%. In the era of nuclear deterrence, large quantities of lithium were processed to remove 6Li for use in hydrogen bomb production. The lithium metal remaining after removal of 6Li was sold on the market. (a) What are the compositions of the nuclei of 6Li and 7Li?
Problem 93b
The nucleus of 6Li is a powerful absorber of neutrons. It exists in the naturally occurring metal to the extent of 7.5%. In the era of nuclear deterrence, large quantities of lithium were processed to remove 6Li for use in hydrogen bomb production. The lithium metal remaining after removal of 6Li was sold on the market. (b) The atomic masses of 6Li and 7Li are 6.015122 and 7.016004 u, respectively. A sample of lithium depleted in the lighter isotope was found on analysis to contain 1.442% 6Li. What is the average atomic weight of this sample of the metal?
- Describe the similarities and differences between the three isotopes of argon, which have 18, 20, and 22 neutrons in the nucleus, respectively.
Problem 94
Problem 94a
The element argon has three naturally occurring isotopes, with 18, 20, and 22 neutrons in the nucleus, respectively. (a) Write the full chemical symbols for these three isotopes.
Problem 95
The element chromium (Cr) consists of four naturally occurring isotopes with atomic masses 49.9460, 51.9405, 52.9407, and 53.9389 u. The relative abundances of these four isotopes are 4.3, 83.8, 9.5, and 2.4%, respectively. From these data, calculate the atomic weight of chromium.
Problem 96
Copper (Cu) consists of two naturally occurring isotopes with masses of 62.9296 and 64.9278 u. (a) How many protons and neutrons are in the nucleus of each isotope? Write the complete atomic symbol for each, showing the atomic number and mass number. (b) The average atomic mass of Cu is 63.55 u. Calculate the abundance of each isotope.
- Using a suitable reference such as the CRC Handbook of Chemistry and Physics or http://www.webelements.com, look up the following information for nickel: (b) the atomic masses (in u), (c) the natural abundances of the five most abundant isotopes.
Problem 97
Problem 98a
There are two different isotopes of bromine atoms. Under normal conditions, elemental bromine consists of Br2 molecules, and the mass of a Br2 molecule is the sum of the masses of the two atoms in the molecule. The mass spectrum of Br2 consists of three peaks: Mass (u) Relative Size 157.836 0.2569 159.834 0.4999 161.832 0.2431 (a) What is the origin of each peak (of what isotopes does each consist)?
Problem 98b
There are two different isotopes of bromine atoms. Under normal conditions, elemental bromine consists of Br2 molecules, and the mass of a Br2 molecule is the sum of the masses of the two atoms in the molecule. The mass spectrum of Br2 consists of three peaks: Mass (u) Relative Size 157.836 0.2569 159.834 0.4999 161.832 0.2431 (b) What is the mass of each isotope?
Problem 98c
There are two different isotopes of bromine atoms. Under normal conditions, elemental bromine consists of Br2 molecules, and the mass of a Br2 molecule is the sum of the masses of the two atoms in the molecule. The mass spectrum of Br2 consists of three peaks: Mass (u) Relative Size 157.836 0.2569 159.834 0.4999 161.832 0.2431 (c) Determine the average molecular mass of a Br2 molecule.
Problem 98d
There are two different isotopes of bromine atoms. Under normal conditions, elemental bromine consists of Br2 molecules, and the mass of a Br2 molecule is the sum of the masses of the two atoms in the molecule. The mass spectrum of Br2 consists of three peaks: Mass (u) Relative Size 157.836 0.2569 159.834 0.4999 161.832 0.2431 (d) Determine the average atomic mass of a bromine atom
Problem 98e
There are two different isotopes of bromine atoms. Under normal conditions, elemental bromine consists of Br2 molecules, and the mass of a Br2 molecule is the sum of the masses of the two atoms in the molecule. The mass spectrum of Br2 consists of three peaks: Mass (u) Relative Size 157.836 0.2569 159.834 0.4999 161.832 0.2431 (e) Calculate the abundances of the two isotopes. Calculate the abundance of the heavier isotope.
