Textbook Question
Look at the location of elements A, B, C, and D in the following periodic table:
(a) Write the formula of the simplest binary hydride of each element.

 Verified step by step guidance
Verified step by step guidance

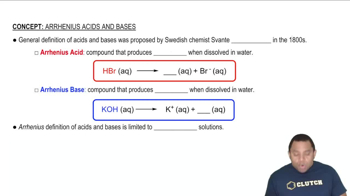
Look at the location of elements A, B, C, and D in the following periodic table:
(a) Write the formula of the simplest binary hydride of each element.
Look at the location of elements A, B, C, and D in the following
periodic table:
(c) Which hydrides react with water to give H2 gas? Write a balanced net ionic equation for each reaction.
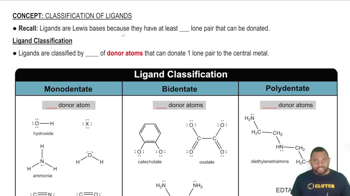
The following models represent the structures of binary hydrides
of second-row elements:
(a) Identify the nonhydrogen atom in each case, and write the molecular formula for each hydride.
Which of the following oxides will be more soluble in acidic solution? (LO 22.14)
(a) SiO2 (b) NO2
(c) BaO (d) B2O3