A drop of water has a volume of approximately 0.05 mL. How many water molecules does it contain? The density of water is 1.0 g/cm3.

Determine the chemical formula of each compound and then use it to calculate the mass percent composition of each constituent element. c. nitrogen triiodide
 Verified step by step guidance
Verified step by step guidance
Verified video answer for a similar problem:
Key Concepts
Chemical Formula
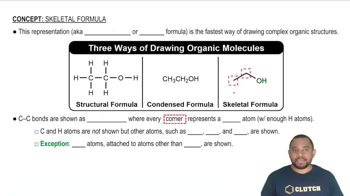
Mass Percent Composition

Molar Mass

Determine the chemical formula of each compound and then use it to calculate the mass percent composition of each constituent element. a. potassium chromate b. lead(II) phosphate
Determine the chemical formula of each compound and then use it to calculate the mass percent composition of each constituent element. c. sulfurous acid
A Freon leak in the air-conditioning system of an old car releases 38.0 g of CF2Cl2 per month. What mass of chlorine does this car emit into the atmosphere each year?
Determine the chemical formula of each compound and then use it to calculate the mass percent composition of each constituent element. d. cobalt(II) bromide
A metal (M) forms a compound with the formula MCl3. If the compound contains 65.57% Cl by mass, what is the identity of the metal?
