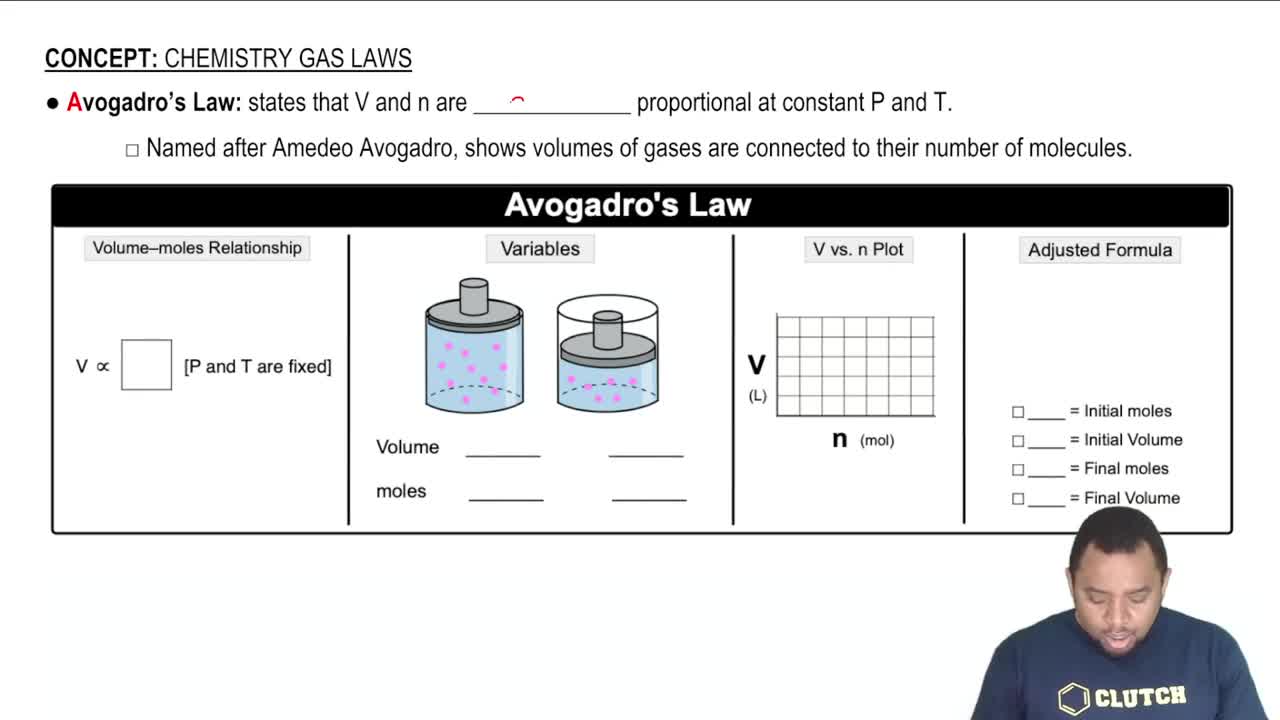
Textbook Question
How many molecules of ethanol (C2H5OH) (the alcohol in alcoholic beverages) are present in 145 mL of ethanol? The density of ethanol is 0.789 g/cm3.
1
views

 Verified step by step guidance
Verified step by step guidance


How many molecules of ethanol (C2H5OH) (the alcohol in alcoholic beverages) are present in 145 mL of ethanol? The density of ethanol is 0.789 g/cm3.
Determine the chemical formula of each compound and then use it to calculate the mass percent composition of each constituent element. a. potassium chromate b. lead(II) phosphate
Determine the chemical formula of each compound and then use it to calculate the mass percent composition of each constituent element. c. sulfurous acid
Determine the chemical formula of each compound and then use it to calculate the mass percent composition of each constituent element. c. nitrogen triiodide