Mothballs are composed of naphthalene, C10H8, a molecule that consists of two six-membered rings of carbon fused along an edge, as shown in this incomplete Lewis structure:(a) Draw all of the resonance structures of naphthalene. How many are there?

(a) The nitrate ion, NO3^-, has a trigonal planar structure with the N atom as the central atom. Draw the Lewis structure(s) for the nitrate ion.
 Verified step by step guidance
Verified step by step guidanceKey Concepts
Lewis Structures

Trigonal Planar Geometry

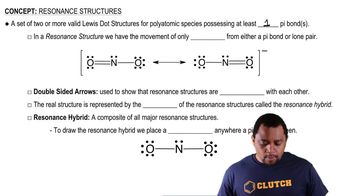
Resonance Structures
Mothballs are composed of naphthalene, C10H8, a molecule that consists of two six-membered rings of carbon fused along an edge, as shown in this incomplete Lewis structure:
(b) Do you expect the C—C bond lengths in the molecule to be similar to those of C—C single bonds, C ═C double bonds, or intermediate between C—C single and C ═C double bonds?
Mothballs are composed of naphthalene, C10H8, a molecule that consists of two six-membered rings of carbon fused along an edge, as shown in this incomplete Lewis structure:
(c) Not all of the C—C bond lengths in naphthalene are equivalent. Based on your resonance structures, how many C—C bonds in the molecule do you expect to be shorter than the others?
a. Triazine, C3H3N3, is like benzene except that in triazine every other C—H group is replaced by a nitrogen atom. Draw the Lewis structure(s) for the triazine molecule.
Ortho-Dichlorobenzene, C6H4Cl2, is obtained when two of the adjacent hydrogen atoms in benzene are replaced with Cl atoms. A skeleton of the molecule is shown here. (a) Complete a Lewis structure for the molecule using bonds and electron pairs as needed.
Ortho-Dichlorobenzene, C6H4Cl2, is obtained when two of the adjacent hydrogen atoms in benzene are replaced with Cl atoms. A skeleton of the molecule is shown here. (b) Are there any resonance structures for the molecule? If so, sketch them.
