(b) Calculate the pH of a buffer formed by mixing 85 mL of 0.13 M lactic acid with 95 mL of 0.15 M sodium lactate.

A buffer is prepared by adding 10.0 g of ammonium chloride (NH4Cl) to 250 mL of 1.00 M NH3 solution. (b) Write the complete ionic equation for the reaction that occurs when a few drops of nitric acid are added to the buffer. (c) Write the complete ionic equation for the reaction that occurs when a few drops of potassium hydroxide solution are added to the buffer.
 Verified step by step guidance
Verified step by step guidanceKey Concepts
Buffer Solutions
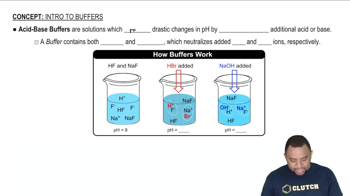
Ionic Equations

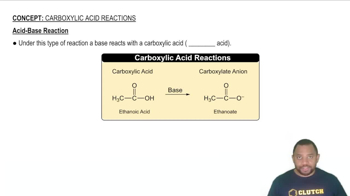
Acid-Base Reactions
You are asked to prepare a pH = 3.00 buffer solution starting from 1.25 L of a 1.00 M solution of hydrofluoric acid (HF) and any amount you need of sodium fluoride (NaF). (a) What is the pH of the hydrofluoric acid solution prior to adding sodium fluoride?
You are asked to prepare a pH = 3.00 buffer solution starting from 1.25 L of a 1.00 M solution of hydrofluoric acid (HF) and any amount you need of sodium fluoride (NaF). (b) How many grams of sodium fluoride should be added to prepare the buffer solution? Neglect the small volume change that occurs when the sodium fluoride is added.
You are asked to prepare a pH = 4.00 buffer starting from 1.50 L of 0.0200 M solution of benzoic acid 1C6H5COOH2 and any amount you need of sodium benzoate 1C6H5COONa2. (a) What is the pH of the benzoic acid solution prior to adding sodium benzoate?
