A sample of nitrosyl bromide (NOBr) decomposes according to the equation 2 NOBr(𝑔) ⇌ 2 NO(𝑔) + Br2(𝑔) An equilibrium mixture in a 5.00-L vessel at 100°C contains 3.22 g of NOBr, 2.46 g of NO, and 6.55 g of Br2. (b) What is the total pressure exerted by the mixture of gases?

As shown in Table 15.2, the equilibrium constant for the reaction N2(g) + 3 H2(g) ⇌ 2 NH3(g) is Kp = 4.34 × 10-3 at 300°C. Pure NH3 is placed in a 1.00-L flask and allowed to reach equilibrium at this temperature. There are 1.05 g NH3 in the equilibrium mixture. (b) What was the initial mass of ammonia placed in the vessel?
 Verified step by step guidance
Verified step by step guidance
Verified video answer for a similar problem:
Key Concepts
Equilibrium Constant (Kp)
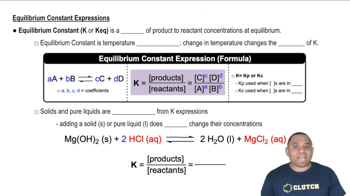
Stoichiometry

Molar Mass and Mass-Volume Relationships

A sample of nitrosyl bromide (NOBr) decomposes according to the equation 2 NOBr(𝑔) ⇌ 2 NO(𝑔) + Br2(𝑔) An equilibrium mixture in a 5.00-L vessel at 100°C contains 3.22 g of NOBr, 2.46 g of NO, and 6.55 g of Br2. (c) What was the mass of the original sample of NOBr?
Consider the hypothetical reaction A(𝑔) ⇌ 2 B(𝑔). A flask is charged with 0.75 atm of pure A, after which it is allowed to reach equilibrium at 0°C. At equilibrium, the partial pressure of A is 0.36 atm. (c) To maximize the yield of product B, would you make the reaction flask larger or smaller?
For the equilibrium PH3BCl3(𝑠) ⇌ PH3(𝑔) + BCl3(𝑔) 𝐾𝑝 = 0.052 at 60°C. (b) A closed 1.500-L vessel at 60°C is charged with 0.0500 g of BCl3(𝑔); 3.00 g of solid PH3BCl3 is then added to the flask, and the system is allowed to equilibrate. What is the equilibrium concentration of PH3?
