Textbook Question
(b) X-ray diffraction studies of buckminsterfullerene show that it has a face-centered cubic lattice of C60 molecules. The length of an edge of the unit cell is 14.2 Å. Calculate the density of buckminsterfullerene.


 Verified step by step guidance
Verified step by step guidance
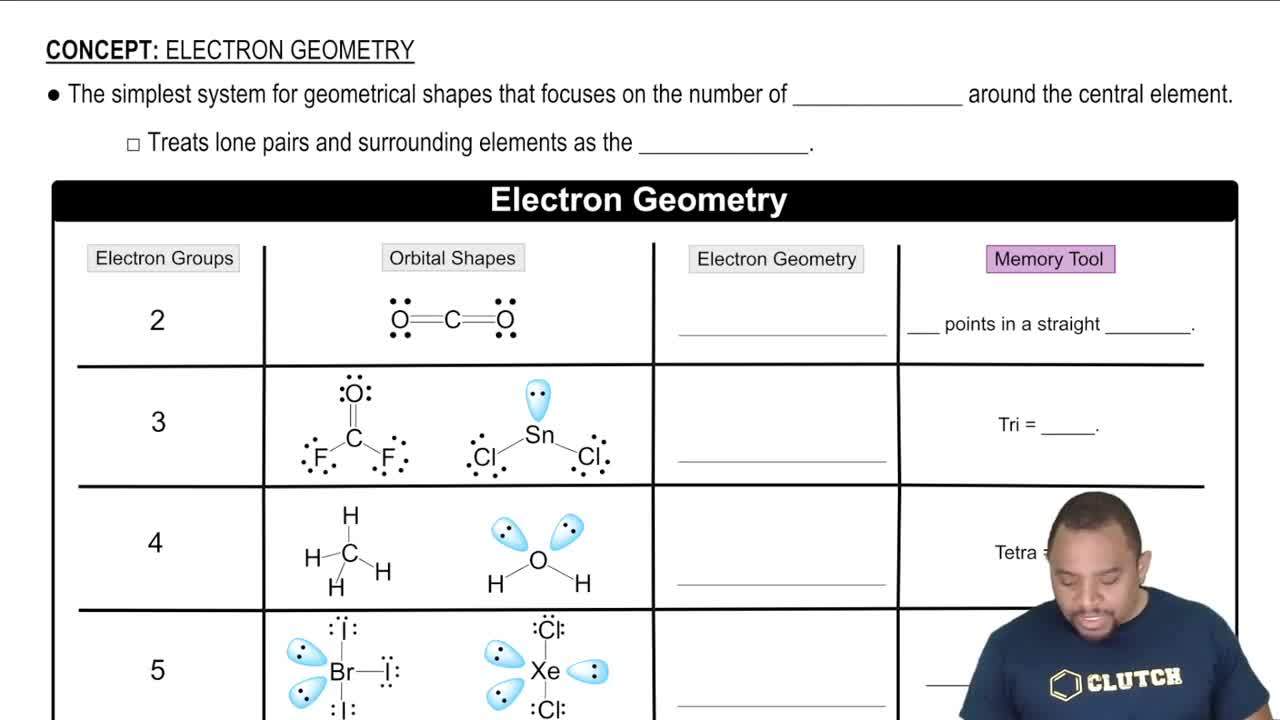

(b) X-ray diffraction studies of buckminsterfullerene show that it has a face-centered cubic lattice of C60 molecules. The length of an edge of the unit cell is 14.2 Å. Calculate the density of buckminsterfullerene.
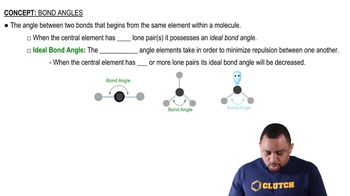
(c) What atomic orbitals are involved in the stacking of graphite sheets with each other?
Employing the bond enthalpy values listed in Table 8.4, estimate the molar enthalpy change occurring upon (a) polymerization of ethylene. (b) formation of nylon 6,6. (c) formation of polyethylene terephthalate (PET).
Employing the bond enthalpy values listed in Table 8.3 estimate the molar enthalpy change occurring upon c. formation of polyethylene terephthalate (PET).