Diethyl ether, C4H10O(l), a flammable compound that was once used as a surgical anesthetic, has the structure The complete combustion of 1 mol of C4H10O(l) to CO2(g) and H2O(l) yields ΔH° = -2723.7 kJ. (a) Write a balanced equation for the combustion of 1 mol of C4H10O(l).

Ethanol (C2H5OH) is blended with gasoline as an automobile fuel. (d) Calculate the mass of CO2 produced per kJ of heat emitted.
 Verified step by step guidance
Verified step by step guidance
Verified video answer for a similar problem:
Key Concepts
Combustion Reaction
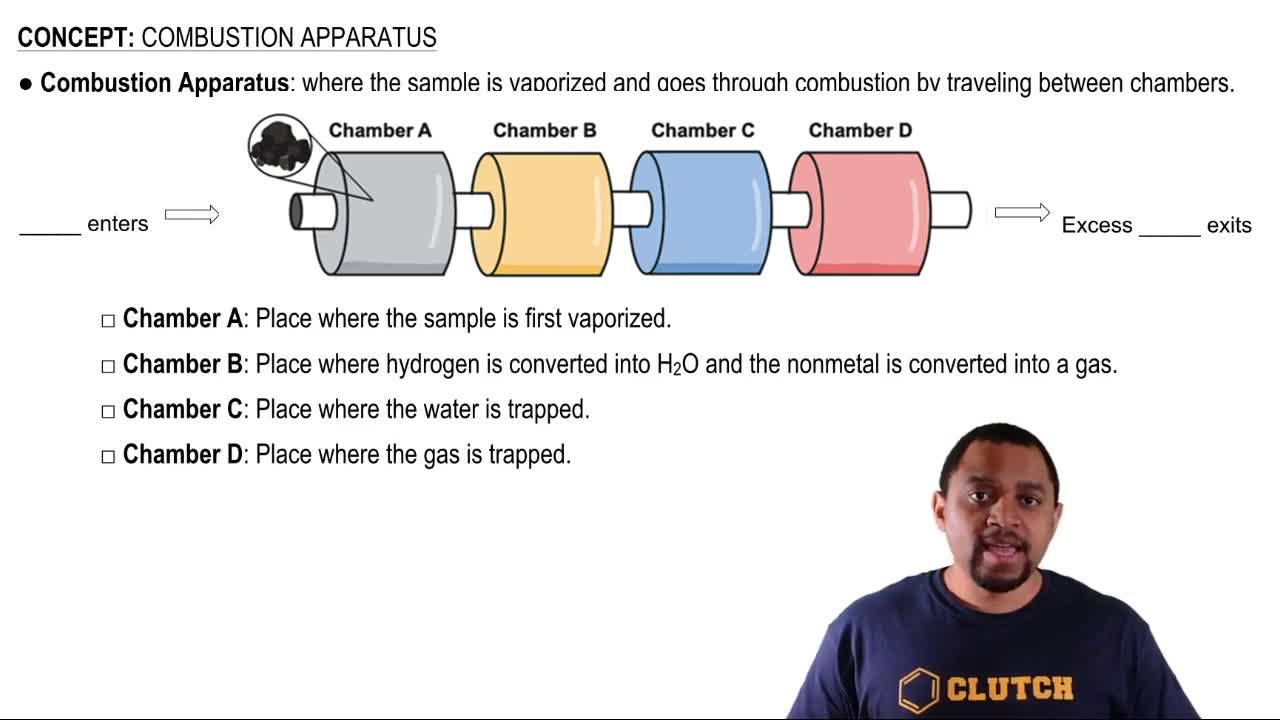
Stoichiometry

Energy Release in Combustion

Ethanol (C2H5OH) is blended with gasoline as an automobile fuel. (b) Calculate the standard enthalpy change for the reaction, assuming H2O(g) as a product.
Ethanol (C2H5OH) is blended with gasoline as an automobile fuel. (c) Calculate the heat produced per liter of ethanol by combustion of ethanol under constant pressure. Ethanol has a density of 0.789 g/mL.
Methanol (CH3OH) is used as a fuel in race cars. (b) Calculate the standard enthalpy change for the reaction, assuming H2O(g) as a product.
Methanol (CH3OH) is used as a fuel in race cars. (c) Calculate the heat produced by combustion per liter of methanol. Methanol has a density of 0.791 g/mL.
Methanol (CH3OH) is used as a fuel in race cars. (d) Calculate the mass of CO2 produced per kJ of heat emitted.
