Derive an equation similar to the Henderson–Hasselbalch equation relating the pOH of a buffer to the pKb of its base component.

The acid–base indicator bromcresol green is a weak acid. The yellow acid and blue base forms of the indicator are present in equal concentrations in a solution when the pH is 4.68. What is the pKa for bromcresol green?
 Verified step by step guidance
Verified step by step guidance
Verified video answer for a similar problem:
Key Concepts
Acid-Base Indicators
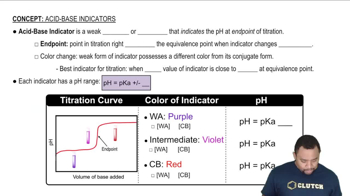
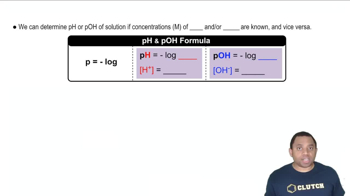
pKa and pH Relationship
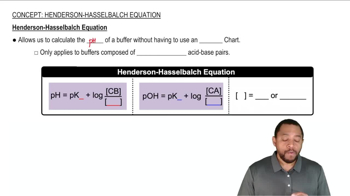
Henderson-Hasselbalch Equation
Rainwater is acidic because CO21g2 dissolves in the water, creating carbonic acid, H2CO3. If the rainwater is too acidic, it will react with limestone and seashells (which are principally made of calcium carbonate, CaCO3). Calculate the concentrations of carbonic acid, bicarbonate ion 1HCO3-2 and carbonate ion 1CO32 - 2 that are in a raindrop that has a pH of 5.60, assuming that the sum of all three species in the raindrop is 1.0 * 10-5 M.
Two buffers are prepared by adding an equal number of moles of formic acid (HCOOH) and sodium formate (HCOONa) to enough water to make 1.00 L of solution. Buffer A is prepared using 1.00 mol each of formic acid and sodium formate. Buffer B is prepared by using 0.010 mol of each. (b) Which buffer will have the greater buffer capacity?
