Suppose that the reaction A¡products is exothermic and has an activation barrier of 75 kJ/mol. Sketch an energy diagram showing the energy of the reaction as a function of the progress of the reaction. Draw a second energy curve showing the effect of a catalyst.

How long will it take for 90% of the CH3CN to convert to CH3NC at 500 °C given the tabulated data: Time (h) [CH3CN] (M) 0.0 1.000, 5.0 0.794, 10.0 0.631, 15.0 0.501, 20.0 0.398, 25.0 0.316?
 Verified step by step guidance
Verified step by step guidanceKey Concepts
Reaction Kinetics
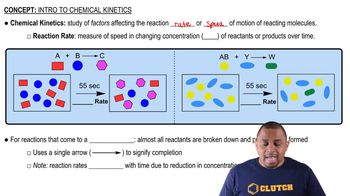
First-Order Reactions

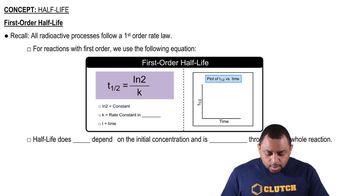
Half-Life of Reactions
The activation barrier for the hydrolysis of sucrose into glucose and fructose is 108 kJ/mol. If an enzyme increases the rate of the hydrolysis reaction by a factor of 1 million, how much lower must the activation barrier be when sucrose is in the active site of the enzyme? (Assume that the frequency factors for the catalyzed and uncatalyzed reactions are identical and a temperature of 25 °C.)
The tabulated data were collected for this reaction at 500 °C: CH3CN(g) → CH3NC( g) a. Determine the order of the reaction and the value of the rate constant at this temperature.
The tabulated data were collected for this reaction at 500 °C: CH3CN(g) → CH3NC( g) b. What is the half-life for this reaction (at the initial concentration)?
The tabulated data were collected for this reaction at a certain temperature: X2Y → 2 X + Y a. Determine the order of the reaction and the value of the rate constant at this temperature.
