The cell potential of this electrochemical cell depends on the gold concentration in the cathode half-cell. Pt(s) | H2(g, 1.0 atm) | H+(aq, 1.0 M) || Au3+(aq, ? M) | Au(s) What is the concentration of Au3+ in the solution if Ecell is 1.22 V?

A battery relies on the oxidation of magnesium and the reduction of Cu2+. The initial concentrations of Mg2+ and Cu2+ are 1.0 × 10–4 M and 1.5 M, respectively, in 1.0-liter half-cells. b. What is the voltage of the battery after delivering 5.0 A for 8.0 h?
 Verified step by step guidance
Verified step by step guidance
Verified video answer for a similar problem:
Key Concepts
Electrochemical Reactions
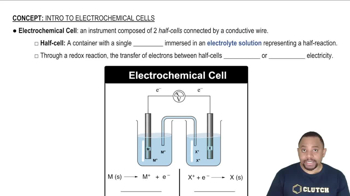
Nernst Equation

Current and Charge

A friend wants you to invest in a new battery she has designed that produces 24 V in a single voltaic cell. Why should you be wary of investing in such a battery?
A battery relies on the oxidation of magnesium and the reduction of Cu2+. The initial concentrations of Mg2+ and Cu2+ are 1.0 × 10–4 M and 1.5 M, respectively, in 1.0-liter half-cells. a. What is the initial voltage of the battery?
A battery relies on the oxidation of magnesium and the reduction of Cu2+. The initial concentrations of Mg2+ and Cu2+ are 1.0 × 10–4 M and 1.5 M, respectively, in 1.0-liter half-cells. c. How long can the battery deliver 5.0 A before going dead?
A rechargeable battery is constructed based on a concentration cell constructed of two Ag/Ag+ half-cells. The volume of each half-cell is 2.0 L, and the concentrations of Ag+ in the half-cells are 1.25 M and 1.0×10–3 M. a. How long can this battery deliver 2.5 Aof current before it goes dead?
A rechargeable battery is constructed based on a concentration cell constructed of two Ag/Ag+ half-cells. The volume of each half-cell is 2.0 L, and the concentrations of Ag+ in the half-cells are 1.25 M and 1.0 × 10–3 M. b. What mass of silver is plated onto the cathode by running at 3.5 A for 5.5 h?
