Methanol (CH3OH) burns in oxygen to form carbon dioxide and water. Write a balanced equation for the combustion of liquid methanol and calculate ΔH°rxn, ΔS°rxn, and ΔG°rxn at 25 °C. Is the combustion of methanol spontaneous?

For each reaction, calculate ΔH°rxn, ΔS°rxn, and ΔG°rxn at 25 °C and state whether the reaction is spontaneous. If the reaction is not spontaneous, would a change in temperature make it spontaneous? If so, should the temperature be raised or lowered from 25 °C?b. NH4Cl(s) → HCl(g) + NH3(g)c. 3 H2(g) + Fe2O3(s) → 2 Fe(s) + 3 H2O(g)
 Verified step by step guidance
Verified step by step guidanceKey Concepts
Thermodynamic Functions (ΔH, ΔS, ΔG)

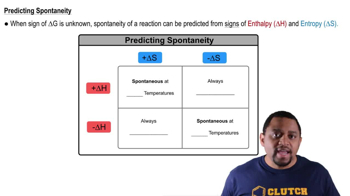
Spontaneity of Reactions

Temperature Effects on Spontaneity
In photosynthesis, plants form glucose (C6H12O6) and oxygen from carbon dioxide and water. Write a balanced equation for photosynthesis and calculate ΔH°rxn, ΔS°rxn, and ΔG°rxn at 25 °C. Is photosynthesis spontaneous?
For each reaction, calculate ΔH°rxn, ΔS°rxn, and ΔG°rxn at 25 °C and state whether or not the reaction is spontaneous. If the reaction is not spontaneous, would a change in temperature make it spontaneous? If so, should the temperature be raised or lowered from 25 °C? a. N2O4(g) → 2 NO2(g)
For each reaction, calculate ΔH°rxn, ΔS°rxn, and ΔG°rxn at 25 °C and state whether or not the reaction is spontaneous. If the reaction is not spontaneous, would a change in temperature make it spontaneous? If so, should the temperature be raised or lowered from 25 °C? d. N2(g) + 3 H2(g) → 2 NH3(g)
