Which of the following complexes can exist as enantiomers? Draw their structures.
(a) [Cr(en)3]3+
(b) cis-[Co(NH3)Cl]2+
(c) trans-[Co(en)2(NH3)Cl]2+
(d) [Pt(NH3)3Cl3]+

 Verified step by step guidance
Verified step by step guidance


Which of the following complexes can exist as enantiomers? Draw their structures.
(a) [Cr(en)3]3+
(b) cis-[Co(NH3)Cl]2+
(c) trans-[Co(en)2(NH3)Cl]2+
(d) [Pt(NH3)3Cl3]+
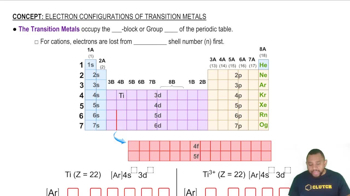
Predict the number of unpaired electrons for each of the following.
(a) Sc3+
(b) Co2+
What is the highest oxidation state for each of the elements from Sc to Zn?
What is the systematic name for each of the following coordination compounds?
(a) Cs[FeCl4]
(b) [V(H2O)6](NO3)3
Nickel(II) complexes with the formula NiX2L2, where X− is Cl− or N-bonded NCS− and L is the monodentate triphenylphosphine ligand P(C6H5)3, can be square planar or tetrahedral.
(a) Draw crystal field energy-level diagrams for a square planar and a tetrahedral nickel(II) complex, and show the population of the orbitals.
Six isomers for a square planar palladium(II) complex that contains two Cl-and two SCN-ligands are shown below.
(a) Which structures are cis-trans isomers?
(b) Which structures are linkage isomers?