Textbook Question
A bag of Hershey's Kisses contains the following information: Serving size: 9 pieces = 41 g Calories per serving: 230 Total fat per serving: 13 g (c) How many Calories are in one Hershey's Kiss?
4
views

 Verified step by step guidance
Verified step by step guidance

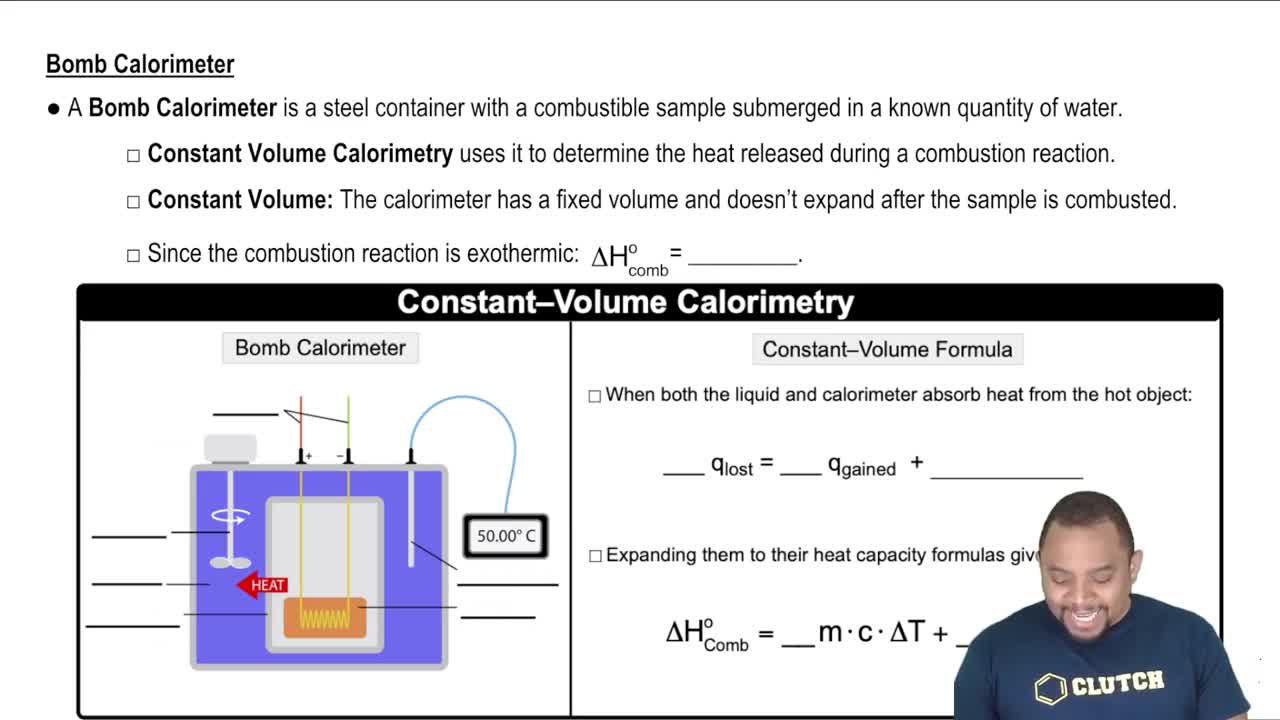
A bag of Hershey's Kisses contains the following information: Serving size: 9 pieces = 41 g Calories per serving: 230 Total fat per serving: 13 g (c) How many Calories are in one Hershey's Kiss?
Ocean currents are measured in Sverdrups (sv) where 1 sv = 109 m3/s. The Gulf Stream off the tip of Florida, for instance, has a flow of 35 sv. (a) What is the flow of the Gulf Stream in milliliters per minute?