The value of Ksp for Cd(OH)2 is 2.5 × 10–14. (b) The solubility of Cd(OH)2 can be increased through formation of the complex ion CdBr42- (Kf = 5 × 103). If solid Cd(OH)2 is added to a NaBr solution, what is the initial concentration of NaBr needed to increase the molar solubility of Cd(OH)2 to 1.0 × 10-3 mol/L?

What is the pH at 25 C of water saturated with CO2 at a partial pressure of 1.10 atm? The Henry's law constant for CO2 at 25 C is 3.1 * 10-2 mol>L@atm.
 Verified step by step guidance
Verified step by step guidance
Verified video answer for a similar problem:
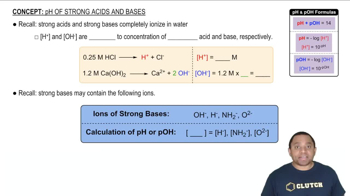
Key Concepts
pH and Acidity
Henry's Law

Carbonic Acid Formation
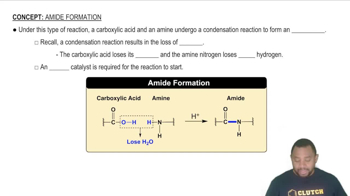
(a) Write the net ionic equation for the reaction that occurs when a solution of hydrochloric acid (HCl) is mixed with a solution of sodium formate 1NaCHO22.
The osmotic pressure of a saturated solution of strontium sulfate at 25 C is 21 torr. What is the solubility product of this salt at 25 C?
A concentration of 10–100 parts per billion (by mass) of Ag+ is an effective disinfectant in swimming pools. However, if the concentration exceeds this range, the Ag+ can cause adverse health effects. One way to maintain an appropriate concentration of Ag+ is to add a slightly soluble salt to the pool. Using Ksp values from Appendix D, calculate the equilibrium concentration of Ag+ in parts per billion that would exist in equilibrium with (b) AgBr (c) AgI.
