Three cations, Ni2+ , Cu2+ , and Ag+, are separated using two different precipitating agents. Based on Figure 17.23, what two precipitating agents could be used? Using these agents, indicate which of the cations is A, which is B, and which is C.[Section 17.7]

Use information from Appendix D to calculate the pH of (a) a solution that is 0.060 M in potassium propionate (C2H5COOK or KC3H5O2) and 0.085 M in propionic acid (C2H5COOH or HC3H5O2). (b) a solution that is 0.075 M in trimethylamine (CH3)3N and 0.10 M in trimethylammonium chloride (CH3)3NHCl. (c) a solution that is made by mixing 50.0 mL of 0.15 M acetic acid and 50.0 mL of 0.20 M sodium acetate.
 Verified step by step guidance
Verified step by step guidanceKey Concepts
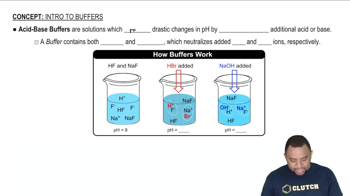
Buffer Solutions
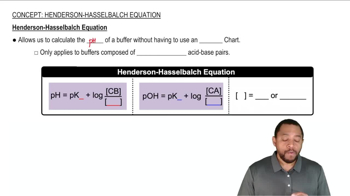
Henderson-Hasselbalch Equation
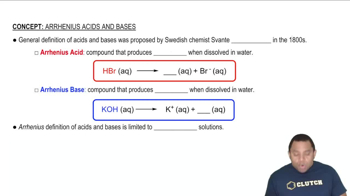
Acid-Base Equilibria
Which of these statements about the common-ion effect is most correct? (a) The solubility of a salt MA is decreased in a solution that already contains either M+ or A-. (b) Common ions alter the equilibrium constant for the reaction of an ionic solid with water. (c) The common-ion effect does not apply to unusual ions like SO32 - . (d) The solubility of a salt MA is affected equally by the addition of either A- or a noncommon ion.
Consider the equilibrium B(aq) + H2O(l) ⇌ HB+(aq) + OH–(aq). Suppose that a salt of HB+(aq) is added to a solution of B(aq) at equilibrium. (c) Will the pH of the solution increase, decrease, or stay the same?
a. Calculate the percent ionization of 0.007 M butanoic acid (𝐾𝑎=1.5×10−5).
(b) Calculate the percent ionization of 0.0075 M butanoic acid in a solution containing 0.085 M sodium butanoate.
