At standard temperature and pressure, the molar volumes of Cl2 and NH3 gases are 22.06 and 22.40 L, respectively. (c) The densities of crystalline Cl2 and NH3 at 160 K are 2.02 and 0.84 g/cm3, respectively. Calculate their molar volumes.

(a) Which type of intermolecular attractive force operates between all molecules?
 Verified step by step guidance
Verified step by step guidance
Verified video answer for a similar problem:
Key Concepts
Intermolecular Forces
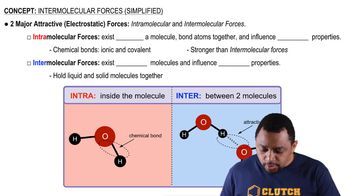
London Dispersion Forces

Polarity and Dipole-Dipole Interactions

At standard temperature and pressure, the molar volumes of Cl2 and NH3 gases are 22.06 and 22.40 L, respectively. (d) Are the molar volumes in the solid state as similar as they are in the gaseous state?
Benzoic acid, C6H5COOH, melts at 122 °C. The density in the liquid state at 130 °C is 1.08 g/cm3. The density of solid benzoic acid at 15 °C is 1.266 g/cm3. (b) If you converted a cubic centimeter of liquid benzoic acid into a solid, would the solid take up more, or less, volume than the original cubic centimeter of liquid?
(b) Which type of intermolecular attractive force operates only between polar molecules?
(c) Which type of intermolecular attractive force operates only between the hydrogen atom of a polar bond and a nearby small electronegative atom?
(b) Which of these kinds of interactions are broken when a liquid is converted to a gas?
