A mixture containing KClO3, K2CO3, KHCO3, and KCl was heated, producing CO2, O2, and H2O gases according to the following equations: 2 KClO31s2¡2 KCl1s2 + 3 O21g2 2 KHCO31s2¡K2O1s2 + H2O1g2 + 2 CO21g2 K2CO31s2¡K2O1s2 + CO21g2 The KCl does not react under the conditions of the reaction. If 100.0 g of the mixture produces 1.80 g of H2O, 13.20 g of CO2, and 4.00 g of O2, what was the composition of the original mixture? (Assume complete decomposition of the mixture.) How many grams of K2CO3 were in the original mixture?

Boron nitride, BN, is an electrical insulator with remarkable thermal and chemical stability. Its density is 2.1 g/cm3. It can be made by reacting boric acid, H3BO3, with ammonia. The other product of the reaction is water. (b) If you made 225 g of boric acid react with 150 g of ammonia, what mass of BN could you make? (d) One application of BN is as a thin film for electrical insulation. If you take the mass of BN from part (b) and make a 0.4 mm thin film from it, what area, in cm2, would it cover?
 Verified step by step guidance
Verified step by step guidanceKey Concepts
Stoichiometry

Density and Volume Calculations

Area Calculation from Volume
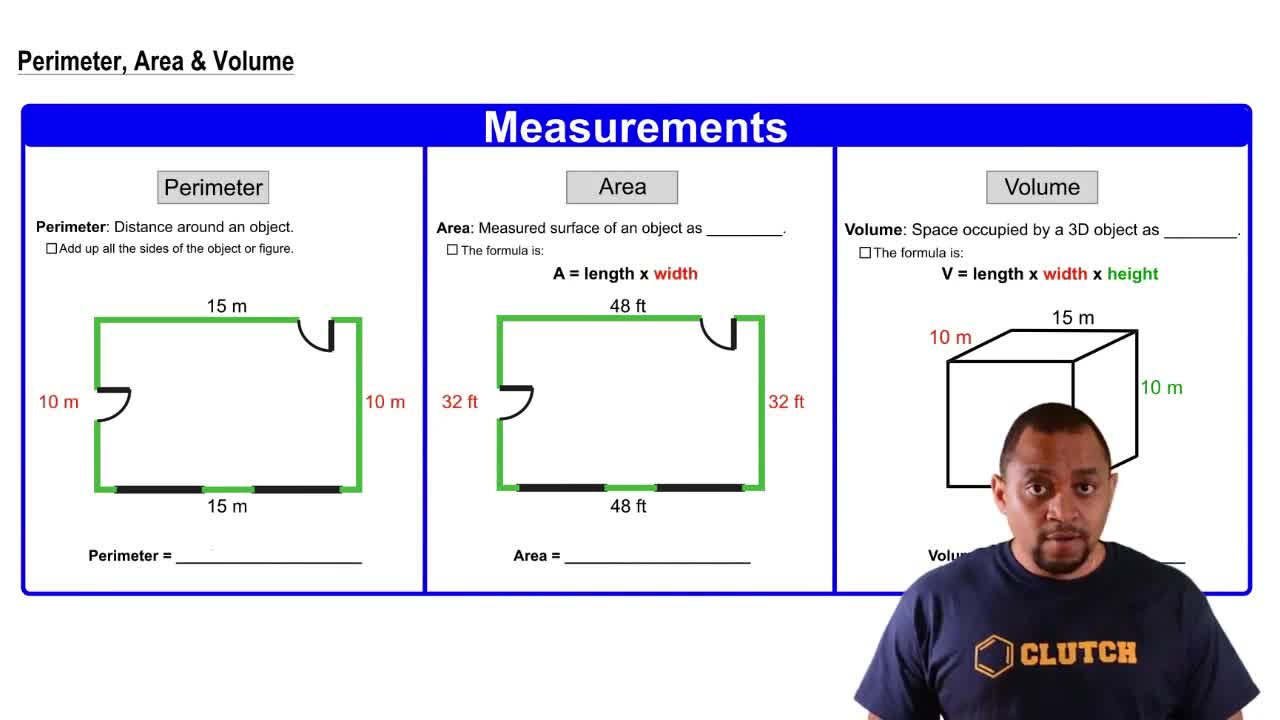
When a mixture of 10.0 g of acetylene (C2H2) and 10.0 g of oxygen (O2) is ignited, the resulting combustion reaction produces CO2 and H2O. (c) How many grams of CO2 and H2O are present after the reaction is complete?
When a mixture of 10.0 g of acetylene (C2H2) and 10.0 g of oxygen (O2) is ignited, the resulting combustion reaction produces CO2 and H2O. (c) How many grams of C2H2 are present after the reaction is complete?
(b) Because atoms are spherical, they cannot occupy all of the space of the cube. The silver atoms pack in the solid in such a way that 74% of the volume of the solid is actually filled with the silver atoms. Calculate the volume of a single silver atom.
Burning acetylene in oxygen can produce three different carbon-containing products: soot (very fine particles of graphite), CO(g), and CO2(g). (c) Why, when the oxygen supply is adequate, is CO2(g) the predominant carbon-containing product of the combustion of acetylene?
