Textbook Question
A voltaic cell utilizes the following reaction: 4 Fe2+1aq2 + O21g2 + 4 H+1aq2 ¡ 4 Fe3+1aq2 + 2 H2O1l2 (a) What is the emf of this cell under standard conditions?

 Verified step by step guidance
Verified step by step guidance
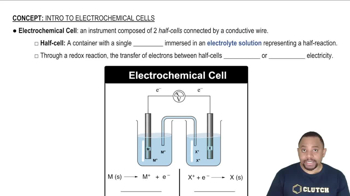


A voltaic cell utilizes the following reaction: 4 Fe2+1aq2 + O21g2 + 4 H+1aq2 ¡ 4 Fe3+1aq2 + 2 H2O1l2 (a) What is the emf of this cell under standard conditions?
A voltaic cell utilizes the following reaction: 4 Fe2+1aq2 + O21g2 + 4 H+1aq2 ¡ 4 Fe3+1aq2 + 2 H2O1l2 (b) What is the emf of this cell when 3Fe2+4 = 1.3 M, 3Fe3+4= 0.010 M, PO2 = 0.50 atm, and the pH of the solution in the cathode half-cell is 3.50?
A voltaic cell utilizes the following reaction: (b) What is the emf for this cell when 3Fe3+4 = 3.50 M, PH2= 0.95 atm, 3Fe2+4 = 0.0010 M, and the pH in both half-cells is 4.00?