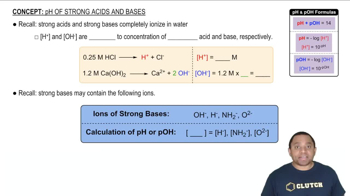
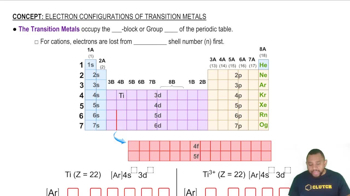
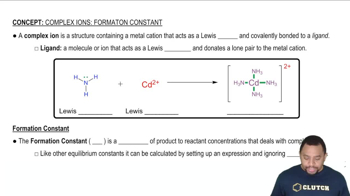
Identify the Lewis acid and Lewis base among the reactants in each of the following reactions:
(a) Fe(ClO4)3(s) + 6 H2O(l) ⇌ [Fe(H2O)6]3+(aq) + 3 ClO4-(aq)
(b) CN-(aq) + H2O(l) ⇌ HCN(aq) + OH-(aq)
(c) (CH3)3N(g) + BF3(g) ⇌ (CH3)NBF3(s)
(d) HIO(lq) + NH2-(lq) ⇌ NH3(lq) + IO-(lq) (lq denotes liquid ammonia as solvent)