Textbook Question
What is the molarity of each of the following solutions: (b) 5.25 g of Mn(NO3)2⋅2H2O in 175 mL of solution,
2
views

 Verified step by step guidance
Verified step by step guidance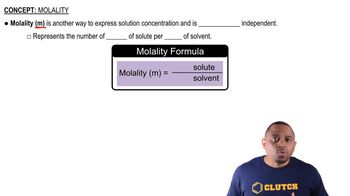


What is the molarity of each of the following solutions: (b) 5.25 g of Mn(NO3)2⋅2H2O in 175 mL of solution,
What is the molarity of each of the following solutions: (c) 35.0 mL of 9.00 M H2SO4 diluted to 0.500 L?
Ascorbic acid (vitamin C, C6H8O6) is a water-soluble vitamin. A solution containing 80.5 g of ascorbic acid dissolved in 210 g of water has a density of 1.22 g/mL at 55 °C. Calculate (a) the mass percentage,
Ascorbic acid (vitamin C, C6H8O6) is a water-soluble vitamin. A solution containing 80.5 g of ascorbic acid dissolved in 210 g of water has a density of 1.22 g/mL at 55 °C. Calculate (c) the molality,