Textbook Question
Introduction of carbon into a metallic lattice generally results in a harder, less ductile substance with lower electrical and thermal conductivities. Explain why this might be so.

 Verified step by step guidance
Verified step by step guidance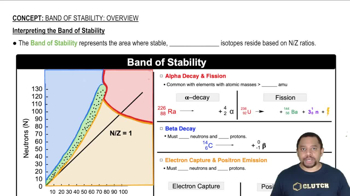

What type of lattice—primitive cubic, body-centered cubic, or face-centered cubic—does each of the following structure types possess: (e) ZnS?
Silicon carbide, SiC, has the three-dimensional structure shown in the figure.
(b) Would you expect the bonding in SiC to be predominantly ionic, metallic, or covalent?