Write the electron configurations for the following ions, and determine which have noble-gas configurations.
a. Ru3+
b. As3−
c. Y3+
d. Pd2+
e. Pb2+
f. Au3+

 Verified step by step guidance
Verified step by step guidance

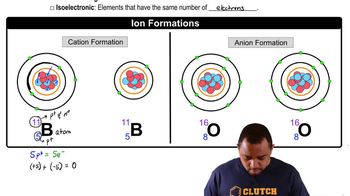
Write the electron configurations for the following ions, and determine which have noble-gas configurations.
a. Ru3+
b. As3−
c. Y3+
d. Pd2+
e. Pb2+
f. Au3+
Which of the ions Ni2+, Fe2+, Co3+, and Pt2+ has an electron configuration of 𝑛𝑑6(𝑛=3,4,5,…)?
a. Ni2+
b. Fe2+
c. Co3+
d. Pt2+
e. More than one of these
rue or false: If the electron affinity for an element is a negative number, then the anion of the element is more stable than the neutral atom.
Write equations that show the processes that describe the first, second, and third ionization energies of an aluminum atom. Which process would require the least amount of energy?
Based on their positions in the periodic table, predict which atom of the following pairs will have the smaller first ionization energy: (a) Br, Kr (b) C, Ca (c) Li, Rb (d) S, Ge (e) Al, B.