The estimated average concentration of NO2 in air in the United States in 2006 was 0.016 ppm. (a) Calculate the partial pressure of the NO2 in a sample of this air when the atmospheric pressure is 755 torr (99.1 kPa).

The water supply for a midwestern city contains the following impurities: coarse sand, finely divided particulates, nitrate ions, trihalomethanes, dissolved phosphorus in the form of phosphates, potentially harmful bacterial strains, dissolved organic substances. Which of the following processes or agents, if any, is effective in removing each of these impurities: coarse sand filtration, activated carbon filtration, aeration, ozonization, precipitation with aluminum hydroxide?
 Verified step by step guidance
Verified step by step guidance
Verified video answer for a similar problem:
Key Concepts
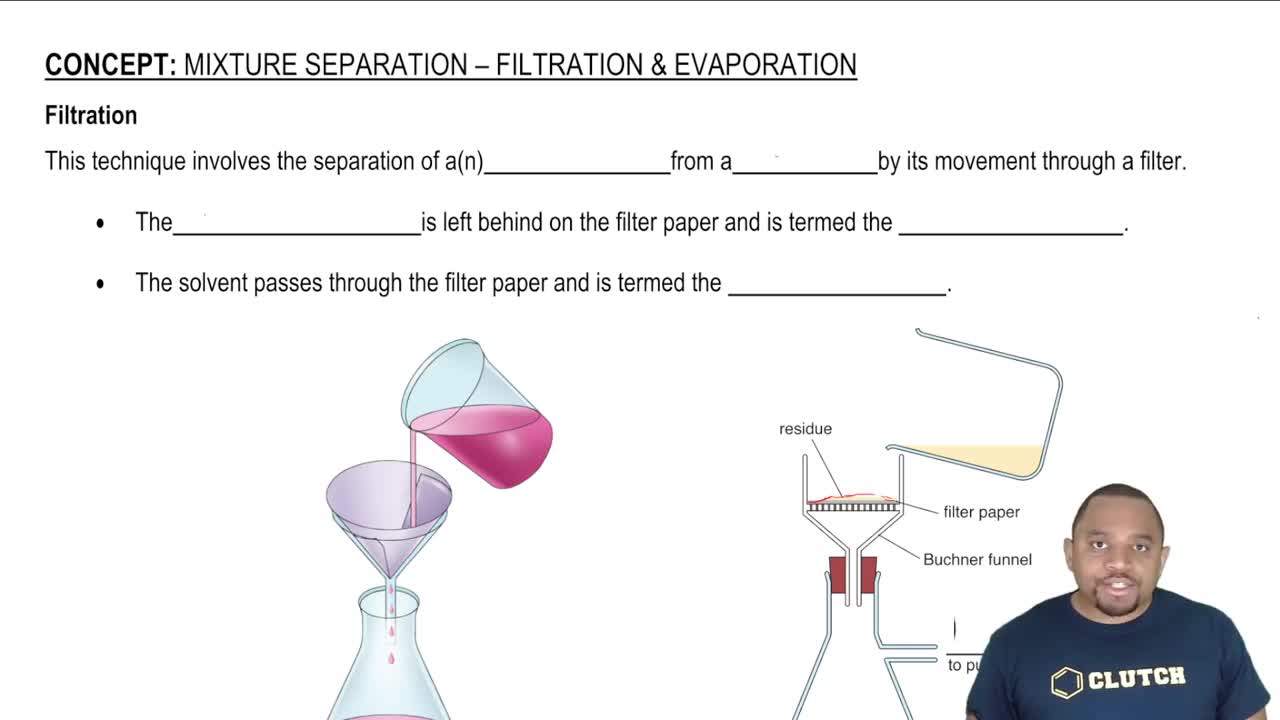
Filtration
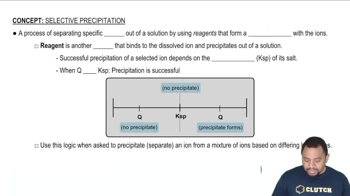
Chemical Precipitation
Ozonization and Aeration

In 1986 an electrical power plant in Taylorsville, Georgia, burned 8,376,726 tons of coal, a national record at that time. (a) Assuming that the coal was 83% carbon and 2.5% sulfur and that combustion was complete, calculate the number of tons of carbon dioxide and sulfur dioxide produced by the plant during the year.
In 1986 an electrical power plant in Taylorsville, Georgia, burned 8,376,726 tons of coal, a national record at that time. (b) If 55% of the SO2 could be removed by reaction with powdered CaO to form CaSO3, how many tons of CaSO3 would be produced?
An impurity in water has an extinction coefficient of 3.45⨉103 M-1 cm-1 at 280 nm, its absorption maximum (A Closer Look, p. 576). Below 50 ppb, the impurity is not a problem for human health. Given that most spectrometers cannot detect absorbances less than 0.0001 with good reliability, is measuring the absorbance of a water sample at 280 nm a good way to detect concentrations of the impurity above the 50-ppb threshold?
The concentration of H2O in the stratosphere is about 5 ppm. It undergoes photodissociation according to: H2O(g) → H(g) + OH(g) (b) Using Table 8.3, calculate the wavelength required to cause this dissociation.
The concentration of H2O in the stratosphere is about 5 ppm. It undergoes photodissociation according to: H2O(g) → H(g) + OH(g)
(c) The hydroxyl radical, OH, can react with ozone, giving the following reactions:
OH(g) + O3(g) → HO2(g) + O2(g)
HO2(g) + O(g) → OH(g) + O2(g)
What overall reaction results from these two elementary reactions? What is the catalyst in the overall reaction? Explain.
