Energy is required to remove two electrons from Ca to form Ca2+, and energy is required to add two electrons to O to form O2 - . Yet CaO is stable relative to the free elements. Which statement is the best explanation? (a) The lattice energy of CaO is large enough to overcome these processes. (b) CaO is a covalent compound, and these processes are irrelevant. (c) CaO has a higher molar mass than either Ca or O. (d) The enthalpy of formation of CaO is small. (e) CaO is stable to atmospheric conditions.

List the individual steps used in constructing a Born–Haber cycle for the formation of BaI2 from the elements. Which of the steps would you expect to be exothermic?
 Verified step by step guidance
Verified step by step guidance
Verified video answer for a similar problem:
Key Concepts
Born-Haber Cycle
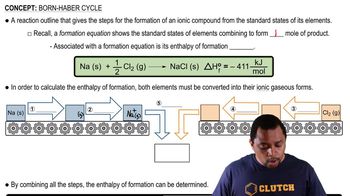
Lattice Energy

Enthalpy Changes

Consider the ionic compounds KF, NaCl, NaBr, and LiCl. (b) Based on your answer to part (a), arrange these four compounds in order of decreasing lattice energy. (c) Check your predictions in part (b) with the experimental values of lattice energy from Table 8.1. Are the predictions from ionic radii correct?
Which of the following trends in lattice energy is due to differences in ionic radii? (a) LiF > NaF > CsF, (b) CaO > KCl, (c) PbS > Li2O.
(a) Based on the lattice energies of MgCl2 and SrCl2 given in Table 8.1, what is the range of values that you would expect for the lattice energy of CaCl2?
(b) Using data from Appendix C, Figure 7.11, Figure 7.13, and the value of the second ionization energy for Ca, 1145 kJ/mol, calculate the lattice energy of CaCl2.
(a) State whether or not the bonding in each substance is likely to be covalent: (i) glucose, (ii) nitrogen, (iii) aluminum hydroxide, (iv) ammonia, (v) neon.
