In ozone, O3, the two oxygen atoms on the ends of the molecule are equivalent to one another. (d) How many electrons are delocalized in the p system of ozone?

Butadiene, C4H6, is a planar molecule that has the following carbon–carbon bond lengths:
(c) The middle C¬C bond length in butadiene (1.48 Å) is a little shorter than the average C¬C single bond length (1.54 Å). Does this imply that the middle C¬C bond in butadiene is weaker or stronger than the average C¬C single bond?
 Verified step by step guidance
Verified step by step guidance
Verified video answer for a similar problem:
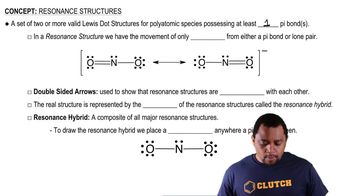
Key Concepts
Bond Length and Bond Strength

Resonance in Molecules
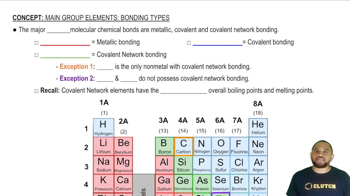
Comparison of Bond Types
Butadiene, C4H6, is a planar molecule that has the following carbon–carbon bond lengths:
(a) Predict the bond angles around each of the carbon atoms and sketch the molecule.
Butadiene, C4H6, is a planar molecule that has the following carbon–carbon bond lengths:
(b) From left to right, what is the hybridization of each carbon atom in butadiene?
The structure of borazine, B3N3H6, is a six-membered ring of alternating B and N atoms. There is one H atom bonded to each B and to each N atom. The molecule is planar. (a) Write a Lewis structure for borazine in which the formal charge on every atom is zero.
The structure of borazine, B3N3H6, is a six-membered ring of alternating B and N atoms. There is one H atom bonded to each B and to each N atom. The molecule is planar. (c) What are the formal charges on the atoms in the Lewis structure from part (b)? Given the electronegativities of B and N, do the formal charges seem favorable or unfavorable? What are the formal charges on the atoms in the Lewis structure from part (b)?
The structure of borazine, B3N3H6, is a six-membered ring of alternating B and N atoms. There is one H atom bonded to each B and to each N atom. The molecule is planar. (e) What are the hybridizations at the B and N atoms in the Lewis structures from parts (a) and (b)? Would you expect the molecule to be planar for both Lewis structures? Would you expect the molecule to be planar for both Lewis structures?
