Textbook Question
The ΔH for the oxidation of sulfur in the gas phase to SO3 is –204 kJ/mol and for the oxidation of SO2 to SO3 is 89.5 kJ/mol. Find the enthalpy of formation of SO2 under these conditions.

 Verified step by step guidance
Verified step by step guidance

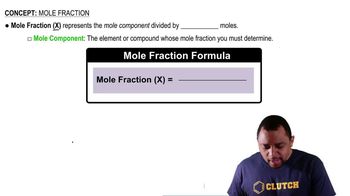
The ΔH for the oxidation of sulfur in the gas phase to SO3 is –204 kJ/mol and for the oxidation of SO2 to SO3 is 89.5 kJ/mol. Find the enthalpy of formation of SO2 under these conditions.
The ΔH°f of TiI3(s) is –328 kJ/mol and the ΔH ° for the reaction 2 Ti(s) + 3 I2(g) → 2 TiI3(s) is –839 kJ. Calculate the ΔH of sublimation of I2(s), which is a solid at 25 °C.
A gaseous fuel mixture contains 25.3% methane (CH4), 38.2% ethane (C2H6), and the rest propane (C3H8) by volume. When the fuel mixture contained in a 1.55 L tank, stored at 755 mmHg and 298 K, undergoes complete combustion, how much heat is emitted? (Assume that the water produced by the combustion is in the gaseous state.)