Textbook Question
Balance each redox reaction occurring in acidic aqueous solution. b. Mg(s) + Cr3+(aq) → Mg2+(aq) + Cr(s)

 Verified step by step guidance
Verified step by step guidance
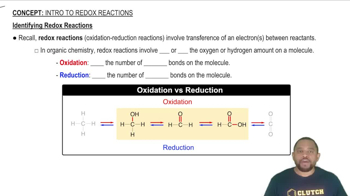


Balance each redox reaction occurring in acidic aqueous solution. b. Mg(s) + Cr3+(aq) → Mg2+(aq) + Cr(s)
Balance each redox reaction occurring in acidic aqueous solution. c. MnO4–(aq) + Al(s) → Mn2+(aq) + Al3+(aq)
Balance each redox reaction occurring in acidic aqueous solution. a. I–(aq) + NO2–(aq) → I2(s) + NO(g) b. ClO4–(aq) + Cl–(aq) → ClO3–(aq) + Cl2(g)
Balance each redox reaction occurring in basic aqueous solution. a. H2O2(aq) + ClO2(aq) → ClO2–(aq) + O2(g)
Balance each redox reaction occurring in basic aqueous solution. b. Al(s) + MnO4–(aq) → MnO2(s) + Al(OH)4–(aq)
Balance each redox reaction occurring in basic aqueous solution. a. MnO4–(aq) + Br–(aq) → MnO2(s) + BrO3–(aq)