Textbook Question
Use tabulated electrode potentials to calculate ∆G°rxn for each reaction at 25 °C. a. 2 Fe3+(aq) + 3 Sn(s) → 2 Fe(s) + 3 Sn2+(aq) b. O2(g) + 2 H2O(l) + 2 Cu(s) → 4 OH–(aq) + 2 Cu2+(aq) c. Br2(l) + 2 I–(aq) → 2 Br–(aq) + I2(s)

 Verified step by step guidance
Verified step by step guidance

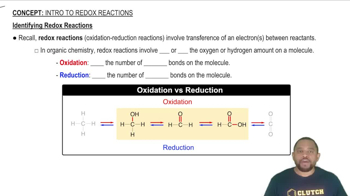

Use tabulated electrode potentials to calculate ∆G°rxn for each reaction at 25 °C. a. 2 Fe3+(aq) + 3 Sn(s) → 2 Fe(s) + 3 Sn2+(aq) b. O2(g) + 2 H2O(l) + 2 Cu(s) → 4 OH–(aq) + 2 Cu2+(aq) c. Br2(l) + 2 I–(aq) → 2 Br–(aq) + I2(s)
Calculate the equilibrium constant for each of the reactions in Problem 65.
A voltaic cell employs the following redox reaction: Sn2+(aq) + Mn(s) → Sn(s) + Mn2+(aq) Calculate the cell potential at 25 °C under each set of conditions. c. [Sn2+] = 2.00 M; [Mn2+] = 0.0100 M