Indicate and explain the sign of ΔSuniv for each process. a. 2 H2(g) + O2(g) → 2 H2O (l) at 298 K.

Is the sign of ΔSuniv for each process positive or negative? Explain for the following: b. the electrolysis of H2O(l) to H2(g) and O2(g) at 298 K c. the growth of an oak tree from a little acorn.
 Verified step by step guidance
Verified step by step guidanceKey Concepts
Entropy (ΔS)
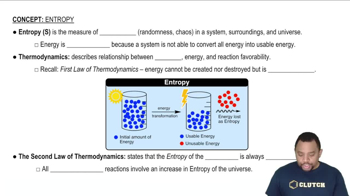
Second Law of Thermodynamics

Spontaneity of Processes

All the oxides of nitrogen have positive values of ΔG°f at 298 K, but only one common oxide of nitrogen has a positive ΔS°f. Identify that oxide of nitrogen without reference to thermodynamic data and explain.
The values of ΔG°f for the hydrogen halides become less negative with increasing atomic number. The ΔG°f of HI is slightly positive. However, the trend in ΔS°f is to become more positive with increasing atomic number. Explain.
A metal salt with the formula MCl2 crystallizes from water to form a solid with the composition MCl2 • 6 H2O. The equilibrium vapor pressure of water above this solid at 298 K is 18.3 mmHg. What is the value of ΔG for the reaction MCl2 • 6 H2O(s) ⇌ MCl2(s) + 6 H2O(g) when the pressure of water vapor is 18.3 mmHg? When the pressure of water vapor is 760 mmHg?
The solubility of AgCl(s) in water at 25 °C is 1.33⨉10-5 mol/L and its ΔH° of solution is 65.7 kJ/mol. What is its solubility at 50.0 °C?
