Textbook Question
The tabulated data were collected for this reaction at 500 °C: CH3CN(g) → CH3NC( g) a. Determine the order of the reaction and the value of the rate constant at this temperature.

 Verified step by step guidance
Verified step by step guidance

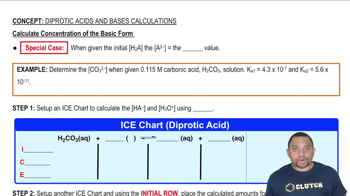
The tabulated data were collected for this reaction at 500 °C: CH3CN(g) → CH3NC( g) a. Determine the order of the reaction and the value of the rate constant at this temperature.
The tabulated data were collected for this reaction at 500 °C: CH3CN(g) → CH3NC( g) b. What is the half-life for this reaction (at the initial concentration)?
The tabulated data were collected for this reaction at a certain temperature: X2Y → 2 X + Y a. Determine the order of the reaction and the value of the rate constant at this temperature.
The tabulated data were collected for this reaction at a certain temperature: X2Y → 2 X + Y c. What is the concentration of X after 10.0 hours?