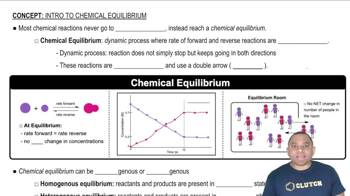
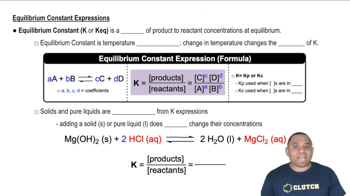
Chemical Equilibrium
Chemical equilibrium occurs when the rates of the forward and reverse reactions are equal, resulting in constant concentrations of reactants and products. At this point, the system is dynamic, meaning that reactions continue to occur, but there is no net change in concentration. Understanding this concept is crucial for writing equilibrium expressions accurately.

 Verified step by step guidance
Verified step by step guidance