Consider the following reaction: 2 NO1g2 + 2 H21g2¡N21g2 + 2 H2O1g2 (d) What is the reaction rate at 1000 K if [NO] is decreased to 0.010 M and 3H24 is increased to 0.030 M?

The reaction between ethyl bromide (C2H5Br) and hydroxide ion in ethyl alcohol at 330 K, C2H5Br(alc) + OH-(alc) → C2H5OH(l) + Br-(alc), is first order each in ethyl bromide and hydroxide ion. When [C2H5Br] is 0.0477 M and [OH-] is 0.100 M, the rate of disappearance of ethyl bromide is 1.7×10-7 M/s. (b) What are the units of the rate constant?
 Verified step by step guidance
Verified step by step guidance
Verified video answer for a similar problem:
Key Concepts
Rate Law

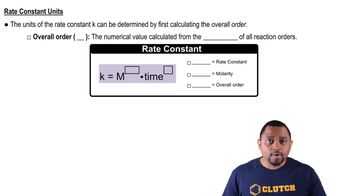
Units of the Rate Constant
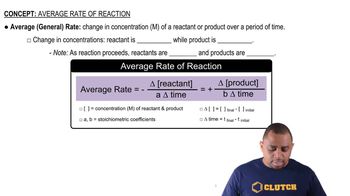
Concentration and Reaction Rate
The reaction between ethyl bromide (C2H5Br) and hydroxide ion in ethyl alcohol at 330 K, C2H5Br(alc) + OH-(alc) → C2H5OH(l) + Br-(alc), is first order each in ethyl bromide and hydroxide ion. When [C2H5Br] is 0.0477 M and [OH-] is 0.100 M, the rate of disappearance of ethyl bromide is 1.7×10-7 M/s. (a) What is the value of the rate constant?
The reaction between ethyl bromide (C2H5Br) and hydroxide ion in ethyl alcohol at 330 K, C2H5Br(alc) + OH-(alc) → C2H5OH(l) + Br-(alc), is first order each in ethyl bromide and hydroxide ion. When [C2H5Br] is 0.0477 M and [OH-] is 0.100 M, the rate of disappearance of ethyl bromide is 1.7×10-7 M/s. (c) How would the rate of disappearance of ethyl bromide change if the solution were diluted by adding an equal volume of pure ethyl alcohol to the solution?
The iodide ion reacts with hypochlorite ion (the active ingredient in chlorine bleaches) in the following way: OCl- + I- → OI- + Cl- . This rapid reaction gives the following rate data:
[OCl4-] (M) [I-] (M) Initial Rate (M,s)
1.5 * 10-3 1.5 * 10-3
1.36 * 10-4 3.0 * 10-3 1.5 * 10-3 2.72 * 10-4
1.5 * 10-3 3.0 * 10-3 2.72 * 10-4
(a) Write the rate law for this reaction.
The iodide ion reacts with hypochlorite ion (the active ingredient in chlorine bleaches) in the following way: OCl - + I - ¡OI - + Cl - . This rapid reaction gives the following rate data:
[OCl4-] (M) [I-] (M) Initial Rate (M,s)
1.5 * 10-3 1.5 * 10-3
1.36 * 10-4 3.0 * 10-3 1.5 * 10-3 2.72 * 10-4
1.5 * 10-3 3.0 * 10-3 2.72 * 10-4
(b) Calculate the rate constant with proper units.
