(a) As described in Section 7.7, the alkali metals react with hydrogen to form hydrides and react with halogens to form halides. Compare the roles of hydrogen and halogens in these reactions. Write balanced equations for the reaction of fluorine with calcium and for the reaction of hydrogen with calcium. (b) What is the oxidation number and electron configuration of calcium in each product?

Compare the elements bromine and chlorine with respect to the following properties: (a) electron configuration (b) most common ionic charge (c) first ionization energy (d) reactivity toward water (e) electron affinity (f) atomic radius
 Verified step by step guidance
Verified step by step guidanceKey Concepts
Electron Configuration
Ionization Energy

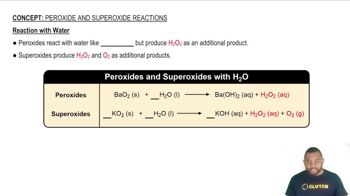
Reactivity with Water
Potassium and hydrogen react to form the ionic compound potassium hydride. (b) Use data in Figures 7.10 and 7.12 to determine the energy change in kJ/mol for the following two reactions:
K(g) + H(g) → K+(g) + H-(g)
K(g) + H(g) → K-(g) + H+(g)
Little is known about the properties of astatine, At, because of its rarity and high radioactivity. Nevertheless, it is possible for us to make many predictions about its properties. (a) Do you expect the element to be a gas, liquid, or solid at room temperature?
Little is known about the properties of astatine, At, because of its rarity and high radioactivity. Nevertheless, it is possible for us to make many predictions about its properties. (b) Would you expect At to be a metal, nonmetal, or metalloid? Explain.
Little is known about the properties of astatine, At, because of its rarity and high radioactivity. Nevertheless, it is possible for us to make many predictions about its properties. (c) What is the chemical formula of the compound it forms with Na?
