Using solubility guidelines, predict whether each of the following compounds is soluble or insoluble in water: (a) MgBr2 (b) NH4OH (c) Ni(CH3COO)2 (d) AgNO3 (e) FeCO3.

Identify the precipitate (if any) that forms when the following solutions are mixed, and write a balanced equation for each reaction. (a) NH4I and CuCl2 (b) LiOH and MnCl2 (c) K3PO4 and CoSO4
 Verified step by step guidance
Verified step by step guidance
Verified video answer for a similar problem:
Key Concepts
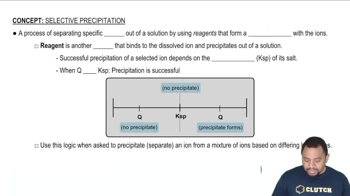
Precipitation Reactions
Solubility Rules
Balanced Chemical Equations

Predict whether each of the following compounds is soluble in water: (a) MgS (b) Cr(OH)3 (c) ZnCl2 (d) Pb3(PO4)2 (e) Sr(CH3COO)2.
Will precipitation occur when the following solutions are mixed? If so, write a balanced chemical equation for the reaction. (a) Ca(CH3COO)2 and NaOH (b) K2CO3 and NH4NO3, (c) Na2S and FeCl3.
Which ions remain in solution, unreacted, after each of the following pairs of solutions is mixed? (a) potassium carbonate and magnesium sulfate
Which ions remain in solution, unreacted, after each of the following pairs of solutions is mixed? (c) ammonium phosphate and calcium chloride
Write balanced net ionic equations for the reactions that occur in each of the following cases. Identify the spectator ion or ions in each reaction.
(a) Ba(OH)2(aq) + FeCl3(aq) →
(b) ZnCl2(aq) + Cs2CO3(aq) →
