Textbook Question
For each of the following pairs, predict which substance possesses the larger entropy per mole: (a) 1 mol of O2(g) at 300 °C, 0.01 atm, or 1 mol of O3(g) at 300 °C, 0.01 atm

 Verified step by step guidance
Verified step by step guidance
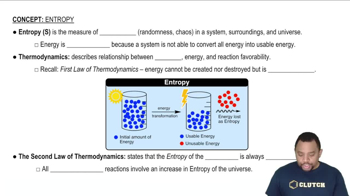


For each of the following pairs, predict which substance possesses the larger entropy per mole: (a) 1 mol of O2(g) at 300 °C, 0.01 atm, or 1 mol of O3(g) at 300 °C, 0.01 atm
Predict the sign of the entropy change of the system for each of the following reactions: (a) N2(g) + 3 H2(g) → 2 NH3(g)
Predict the sign of the entropy change of the system for each of the following reactions: (c) 3 C2H2(g) → C6H6(g)
Predict the sign of the entropy change of the system for each of the following reactions: (d) Al2O3(s) + 3 H2(g) → 2 Al(s) + 3 H2O(g)
Predict the sign of ΔSsys for each of the following processes: (a) Molten gold solidifies.