(a) What atoms must a molecule contain to participate in hydrogen bonding with other molecules of the same kind? (b) Which of the following molecules can form hydrogen bonds with other molecules of the same kind: CH3F, CH3NH2, CH3OH, CH3Br?

Ethylene glycol (HOCH2CH2OH), the major substance in antifreeze, has a normal boiling point of 198 °C. By comparison, ethyl alcohol (CH3CH2OH) boils at 78 °C at atmospheric pressure. Ethylene glycol dimethyl ether (CH3OCH2CH2OCH3) has a normal boiling point of 83 °C, and ethyl methyl ether (CH3CH2OCH3) has a nomral boiling point of 11 °C. (b) What are the major factors responsible for the difference in boiling points of the two ethers?
 Verified step by step guidance
Verified step by step guidance
Verified video answer for a similar problem:
Key Concepts
Intermolecular Forces
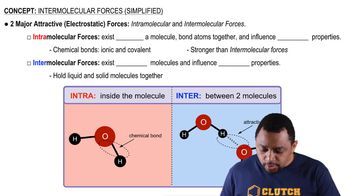
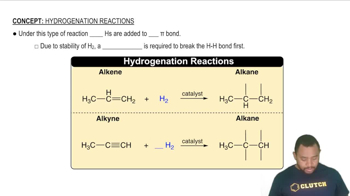
Hydrogen Bonding
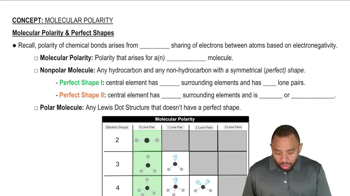
Molecular Structure and Polarity
Ethylene glycol (HOCH2CH2OH), the major substance in antifreeze, has a normal boiling point of 198 °C. By comparison, ethyl alcohol (CH3CH2OH) boils at 78 °C at atmospheric pressure. Ethylene glycol dimethyl ether (CH3OCH2CH2OCH3) has a normal boiling point of 83 °C, and ethyl methyl ether (CH3CH2OCH3) has a nomral boiling point of 11 °C. (a) Explain why replacement of a hydrogen on the oxygen by a CH3 group generally results in a lower boiling point.
Based on the type or types of intermolecular forces, predict the substance in each pair that has the higher boiling point: (a) propane (C3H8) or n-butane (C4H10) (b) diethyl ether (CH3CH2OCH2CH3) or 1-butanol (CH3CH2CH2CH2OH) (c) sulfur dioxide (SO2) or sulfur trioxide (SO3) (d) phosgene (Cl2CO) or formaldehyde (H2CO)
A number of salts containing the tetrahedral polyatomic anion, BF4-, are ionic liquids, whereas salts containing the somewhat larger tetrahedral ion SO42- do not form ionic liquids. Explain this observation.
